
Concept explainers
Compound X
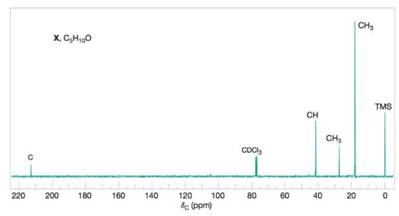
Figure 2 The broadband proton-decoupled
Want to see the full answer?
Check out a sample textbook solution
Chapter FRP Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
Additional Science Textbook Solutions
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Organic Chemistry As a Second Language: Second Semester Topics
Chemistry: A Molecular Approach
Chemistry: Structure and Properties (2nd Edition)
Chemistry: A Molecular Approach (4th Edition)
Introductory Chemistry (6th Edition)
- The 1H and 13C NMR spectra of compound A, C8H9Br, are shown. Propose a structure for A, and assign peaks in the spectra to your structure.arrow_forwardCompound A, a hydrocarbon with M+=96 in its mass spectrum, has the 13C spectral data given below. On reaction with BH3, followed by treatment with basic H2O2. A is converted into B, whose, 13C spectral data are also given below. Propose structures for A and B. Compound A Broadband-decoupled l3C NMR: 26.8, 28.7, 35.7, 106.9, 149.7 DEPT-90: no peaks DEPT-135: no positive peaks; negative peaks at 26.8, 28.7, 35.7, 106.9 Compound B Broadband-decoupled 13C NMR: 26.1, 26.9, 29.9, 40.5, 68.2 DEPT-90: 40.5 DEPT-135: positive peak at 40.5 ; negative peaks at 26.1, 26.9. 29.9, 68.2arrow_forwardH NMR (300 mMHz, CDC13, 21C) 0.92 ppm (t, 3H, J = 7Hz), 1.20 (s, 6H), 1.50 (q, 2H, J = 7 Hz), 1.64 (broad singlet, 1H). Select the correct structure for this molecule. A B OH OH O to o HO Earrow_forward
- ¹H-NMR Spectra 3 M.F. = C₂H₁3NO 1 S 10 -00 8 6 PPM 4 N 21 3 ſ 2[2] -~ 2 3 jarrow_forwardQ4: The proton NMR spectrum of a compound with formula C8H14O2 is shown. The DEPT experimental results are tabulated. The infrared spectrum shows medium-sized bands at 3055, 2960, 2875, and 1660 cm-1 and strong bands at 1725 and 1185 cm-1. Draw the structure of this compound. Normal Carbon DEPT-135 DEPT-90 10.53 ppm Positive No peak Positive No peak No peak No peak 12.03 14.30 Positive 22.14 Negative No peak No peak 65.98 Negative 128.83 No peak 136.73 Positive Positive 168.16 No peak No peak (C=0) 0.97 2.05 7.92 2.96 7.0 6.5 60 5.5 5.0 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0arrow_forwardCompound CsH12 gives the following H-NMR. Draw the structure of the compound. Draw a box around the structure you want graded. 5.00 1.00 6.04 70 6.5 6.0 4.0 3.5 3.0 25 20 1.5 A student was adding bromine across the double bond of 2-butene to make 2,3-dibromobutane. After taking the NMR, the student discovered they didn't get the product expected. Based on the NMR, what product did they obtain? Draw a box around your answer. 1.00 2.01 |2.00 3.00arrow_forward
- PART 2: Compounds A, B, and C are isomers with the formula C5H11Br. Their broadband proton-decoupled 13C NMR spectra are given below. Information from the DEPT 13C NMR spectra is given near each peak. Give structure for C.arrow_forward11 10 9 8 7 9 +6 5 -3 2 1 ppmarrow_forwardWhich C9H100 compound gives the following 1H NMR spectrum? 3H 3H 2H 2H 3. 6. 4 PPM 8. H. 3 4. 1 2.arrow_forward
- 5 Propose structures for compounds whose 1H-NMR spectra and 13C-NMR are given in the attached picture: C6H12arrow_forwardAnalyze the 1H AND 13C NMR spectrum of C8H9NO given below and draw the structure of the compound.arrow_forward11. The proton NMR spectrum for a compound with formula C,HgO is shown below. The normal carbon-13 NMR spectrum has five peaks. The infrared spectrum has a strong band at 1746 cm. The DEPT-135 and DEPT-90 spectral results are tabulated. Draw the structure of this compound. Normal Carbon DEPT-135 DEPT-90 44 ppm Negative No peak ( 125 Positive Positive 127 Positive Positive CA 138 No peak No peak co 215 No peak No peak Proton spectrum C,HgO uluw 0.0 4.10 0.5 4.14 1.0 1.5 2.0 2.5 3.0 3.5 7.5 4.0 70 4.5 6.5 6.0 5.5 5.0arrow_forward
