
Concept explainers
Interpretation:
Reagents used are to be suggested in each part.
Concept introduction:
The reaction which occurs in between two esters or one ester and another carbonyl compound, and forms a carbon–carbon bond, in the presence of a strong base, leading to the formation of a β-keto ester or a β-diketone is known as Claisen condensation.
The removal of carbon dioxide from a carbonyl compound is termed as a decarboxylation reaction.
Sodium borohydride
Hydrogen bromide
Answer to Problem 1PP
Solution:
(a)
Sodium ethoxide
(b)
An acid
(c)
Heat
(d)
Sodium borohydride
(e)
Hydrogen bromide
(f)
Base
Explanation of Solution
The first step is similar to a crossed Claisen condensation. The reagent used in this reaction is sodium ethoxide. The molecular formula of sodium ethoxide is
The reaction shown is as follows:
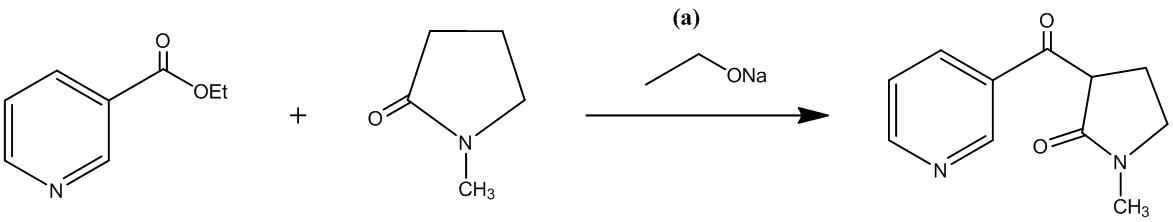
The second step involves the hydrolysis of an amide that is lactamide, which can be carried out with either an acid or a base. In the given reaction the desired product is formed by using an acid.
The reaction is shown as follows:
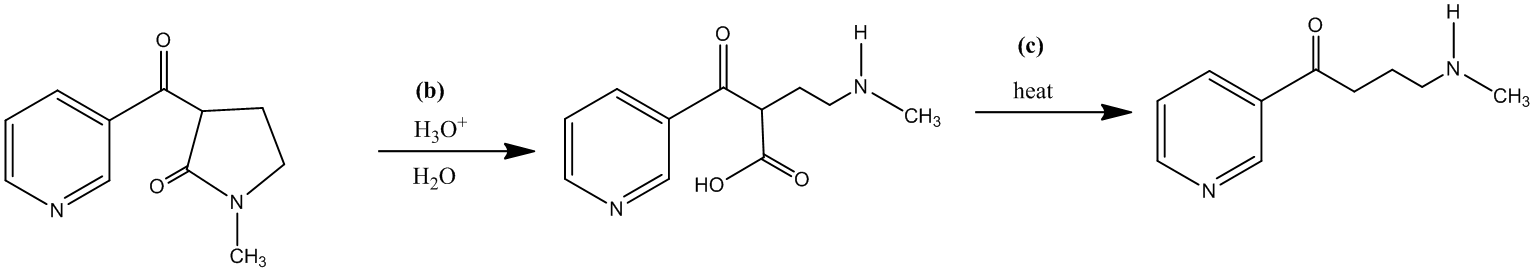
The third step is the decarboxylation of a β-keto acid, which is formed by the acidic hydrolysis of an amide. It requires only the application of heat to get the desired product. The reaction takes place along with the second step.
The forth step is the reduction of the ketone, obtained in third step, into a secondary alcohol. There are many reducing agents, which can reduce a ketone into an alcohol, for instance, sodium borohydride. The molecular formula for the same is
The reaction shown is as follows:
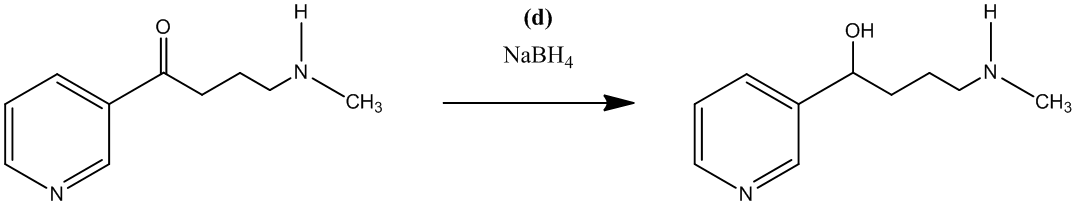
The fifth step is the conversion of the secondary alcohol, obtained in forth step, to an alkyl bromide by using hydrogen bromide. This reagent also gives a hydrobromide salt of the aliphatic
The reaction shown is as follows:
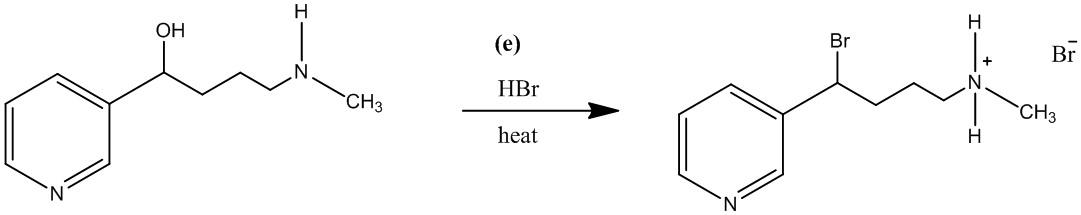
The treatment of the salt with base will lead to the formation of the secondary amine. It will further act as a nucleophile and attack the carbon atom bearing the bromine. This reaction leads to the formation of a five-membered ring and (±) nicotine.
The reaction is shown as follows:
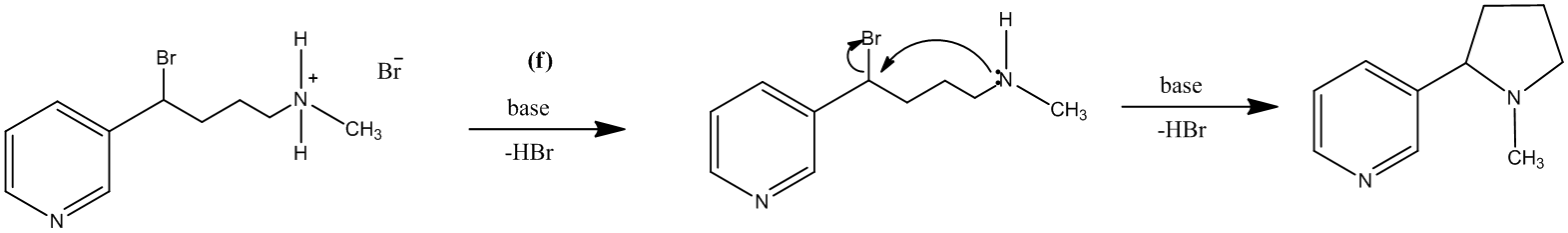
The reagents that could be used for each part are suggested as:
(a): Sodium ethoxide
(b): An acid
(c): Heat
(d): Sodium borohydride
(e): Hydrogen bromide
(f): Base
Want to see more full solutions like this?
Chapter H Solutions
Organic Chemistry, 12e Binder Ready Version Study Guide / Student Solutions Manual
- Organic Chemistry problem. Please help solve. Thank you. Propose a synthesis of cyclobutanecarboxylic acid using diethyl malonate as your ONLY source of carbon atoms. (starting from the starting material on the right side of the picture, how would you arrive at the end product seen on the left side?) Sorry, I know it's confusing looking picture..arrow_forwardWhen cyclohexanone reacts with 2 moles of ethanol and HCl catalyst, cyclohexanone diethyl ketal is produced. Draw a plausible mechanismarrow_forwardFor each reaction below, provide the product(s) that would form and indicate whether the substitution reaction would occur by an SN1 , SN2, E1, or E2 mechanism. Draw all constitutional and/or stereoisomers that would form in each reaction. I need help with this practice problem, thank you!arrow_forward
- Problem 2: Consider the following reaction. Using mechanism and less than 30 words explain the outcome of this reaction.arrow_forwardtrue or false question. ( explanation not needed) a) Gilman copper reagent does 1,2 nucleophilic addition to α,β.unsaturated ketones b)Tertiary alcohol will be formed by the reaction between any ketone and Grignard. c)Cyclopentadiene anion is aromatic d)Tertiary alcohols undergoes dehydration by E1 mechanism e)1,4-addition product is the kinetic product in the addition of conjugated diene f) Benzene is more reactive than toluene in aromatic electrophilic substitution reactionarrow_forwardPractice problem 1: Show the correct major organic products. Thank you !arrow_forward
- What reagents are required to carry out the following reaction (select all that apply)? a HNO3 b H2SO4 c Cl2 d Br2 e H2O f CH3COCl g AlCl3 h FeCl3 i FeBr3 j CH3CHClCH3arrow_forwardIn Problem b and c, which product is the major product. I don’t understand how to find it. Thank you for your helparrow_forwardWhich of the following reaction sequences will produce the product below? Select ALL that apply.arrow_forward
- Predict product belowarrow_forwardThe hydrolysis of CH3-CH=CH-CH2-Cl gives rise to two products: CH3-CH=CH-CH2-OHl and CH3-CH(-OH)CH=CH2. Write a mechanism that accounts for their formation. Show each step clearly. Use curved arrow notation.arrow_forwardWhat is the major product of the following compound. All structure and mechanism I need computer answerarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





