
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
12th Edition
ISBN: 9781119304241
Author: Solomons
Publisher: WILEY C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter H, Problem 7PP
Practice Problem H.7
One of the important steps in Gates’ synthesis of morphine involved the following transformation:
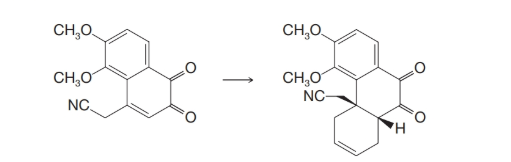
Suggest how this step was accomplished.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Synthesis practice
ork assign
1) Synthesize this compound only using only organic starting materials such as acetic anhydride,
triphenylphosphine, ethyne, and alcohols of four or fewer carbons and any inorganic reagents
needed
CH3
I
CH3CH₂-C-C=CH
I
ОН
PROBLEM 3
Suggest mechanisms for these eliminations. Why does the first give a mixture
and the second a single product?
OH
OH
H₂PO4
heat
X³
OH
48% HBr
heat
64% yield, 4:1 ratio
Problem 2,8
Suggest a plausible arrow-pushing mechanism for the following tautomerization
reactions
cat. HA
OH
cat B
O NH2
NH2
NIH
Chapter H Solutions
ORGANIC CHEM. VOL.1+2-W/WILEYPLUS
Ch. H - Prob. 1PPCh. H - Prob. 2PPCh. H - Prob. 3PPCh. H - Practice Problem H.4
In 1891 G. Merling...Ch. H - Practice Problem H.5
Many alkaloids appear to be...Ch. H - Practice Problem H.6 Papaverine has been...Ch. H - Practice Problem H.7
One of the important steps in...Ch. H - Prob. 8PPCh. H - Practice Problem H.9 Gramine has been synthesized...
Additional Science Textbook Solutions
Find more solutions based on key concepts
APPLY 1.2 Express the following quantities in scientific notation
using fundamental SI units of mass and lengt...
Chemistry (7th Edition)
How many valence electrons are present in the following atoms? a. O b. C c. P d. Na
General, Organic, and Biological Chemistry (3rd Edition)
Q8. Perform the calculation to the correct number of significant figures.
a) 0.121
b) 0.12
c) 0.12131
d) 0.121...
Chemistry: A Molecular Approach (4th Edition)
PRACTICE PROBLEM 19.4
Write mechanisms that account for the products that are formed in the crossed Claisen con...
Organic Chemistry
39. What are the units of k for each type of reaction?
a. first-order reaction
b. second-order reaction
c...
Chemistry: Structure and Properties
a. Draw two Lewis structures for C2H6O. b. Draw three Lewis structures for C3H8O. (Hint: The two Lewis structur...
Essential Organic Chemistry (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify the products of the following reaction. A) H 1) MCPBA 2) H3O+ H3C H3C HO OH H CH3 HO OH B) H&C H H3C CH3 H3C H3C HO OH H CH3 OH D) H3C H H3C CH3 (Select all that apply) U Aarrow_forwardplease help with OChem question Predict the oxidative product(s) of 2-methyl-6-octyn-2-ene after undergoing ozonolysis with O3, H2O2arrow_forwardProblem 3 of 16 Draw the products of this reduction of a ketone with sodium borohydride. 1) NaBH4 49 Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicableIgnore any inorganic byproducts. 2) HCI/H₂O Submit Select to Drawarrow_forward
- OChem Question: Starting with propyne and using any other required reagents, show how you would synthesize (2R, 3R)- and (2S, 3S)-2,3-dibromobutane (as a racemic form)arrow_forwardPROGRESS CHECK 2.4 1. What products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures of all. (a) CH3 Ib) CH3 CH3CHCH=CHCH2CH3 2. How would you carry a out the following trans formations using an organocopper coupling reaction? More than one step is required in each case. (a) CH3 (b) CH3CH2CH2CH2B CH3CH2CH2CH2CH2CH2CH2CH3arrow_forwardWhat reagents are required to carry out the following reaction (select all that apply)? a HNO3 b H2SO4 c Cl2 d Br2 e H2O f CH3COCl g AlCl3 h FeCl3 i FeBr3 j CH3CHClCH3arrow_forward
- Problem 3.3 Draw mechanisms for each of the following substitution reactions. Remember that in each case, loss of the leaving group is preceded by coordination of the leaving group to a Lewis acid such as H+. (a) (b) CH3 CH3 HCl H3C- OH H3C- Cl CH3 CH3 OH ZnCl2 Ar Ar Ar Ar HOarrow_forward36. Which of A - E is the most likely product of the following reaction? [A] Br Mg ether OH [D] 1. 2. 30 [B] OH ? OH (E) [C] LOHarrow_forwardRank these compounds in increasing order of reactivity in an SN2 reaction. Br CI OH II (A) Iarrow_forwardProblem 378 of 36 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. H H H Select to Add Arrows H HCI, H2O H Select to Add Arrowsarrow_forwardProblem 29 of 50 Submit Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic steps. Be sure to account for all bond- breaking and bond-making steps. N Select to Add Arrows THF Na Ⓒ Na O Oarrow_forward23) Which of the following reactions will not produce the given product in good yield? Select all that apply Reagent only reacts once MeONa a) Br MEOH Br Br (CH3),CONa b) Br (CH3)3COH H2N OH 1)(CH3)3CONa 2)arrow_forwardarrow_back_iosSEE MORE QUESTIONSarrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY