10°C 25 °C 37°C Temperature 0 cm CO2 12 cm CO2 20 cm CO2 7 9 pH 11 cm CO2 15 cm CO2 8 cm CO2 2 g 4 g 6 g Yeast 15 cm CO2 22 cm CO2 28 cm CO2 1g 3 g 5 g Sugar 13 cm CO2 17 cm CO2 19 cm CO2
Q: FIGURE 6D CH, — сн,—он NAD* CH-C-H NADH (а) C-OH + C-OH C-OH C-OH C=0 H-C-NH2 H-C-NH2 c=0 CH3 CH3…
A: Proteins that act as a catalyst in the chemical reaction are termed Enzymes. They help in the…
Q: The following questions are based on Figure 6.2 В C A E Prograss af the Reaction Free Energy
A: Answer :: 4) Graph is showing the exergonic reaction, in this graph energy of activation is more in…
Q: An anabolic enzyme:
A: Anabolism involves formation of complex molecule. Gluconeogenesis, glycogenesis, fatty acid…
Q: Label the image. The following image illustrates one of the trials Hershey and Chase used in their…
A: Earlier it was thought that protein was the genetic material which was passed from one generation to…
Q: If ______ alcohol is added to water, the two solutions are ______, forming a ______solution. If…
A: When water is mix with ethanol it form miscible liquids, because any amount of ethanol can be mix…
Q: Which of the following could affect the ability of an enzyme to catalyze a reaction? PH all of these…
A: Note: As Per Guidelines, We Can Answer One Question At A Time. Also you have posted graded questions…
Q: 1. pathway 2. metabolite 3. enzyme 4.reduced coenzyme 5. pathway 6. final product 7.reaction…
A: The series of chemical reactions that occur within the living body are collectively known as…
Q: What is the name of this graph? 100 10 1.0 0.1 60 64 68 72 74 78 Temperature (C) D value (seconds)
A: In pharmaceutical and food industries a unit is defined as sterile, on the statistical basis of…
Q: Without oxygen, 1) pH will increase O2) cells become more basic 3) proteins may change structure
A: PH will increase
Q: Free radicals are highly reactive molecules that often contain ________. They are reactive due to…
A: The free radicals are formed during the metabolic reactions taking place inside the cells. They are…
Q: Endergonic reactions ____. result in products with less energy than the reactants require a net…
A: Endergonic and exergonic reactions are defined according to the change in Gibbs's free energy. In an…
Q: Which of the following best characterizes ANABOLISM? 1.It is largely exergonic reaction because it…
A: The process of metabolism encompasses both anabolism and catabolism.
Q: The 2-carbon alcohol called ethanol is most common because it's made from something else very…
A: Ethanol is chemical compound which have chemical formula C2H6O . Uses of ethanol -- To treat…
Q: the rate of the reaction in one direction is equal to the rate in the reverse direction. the…
A: At equilibrium, reaction appears to have stopped, and the total amount of products and reactants…
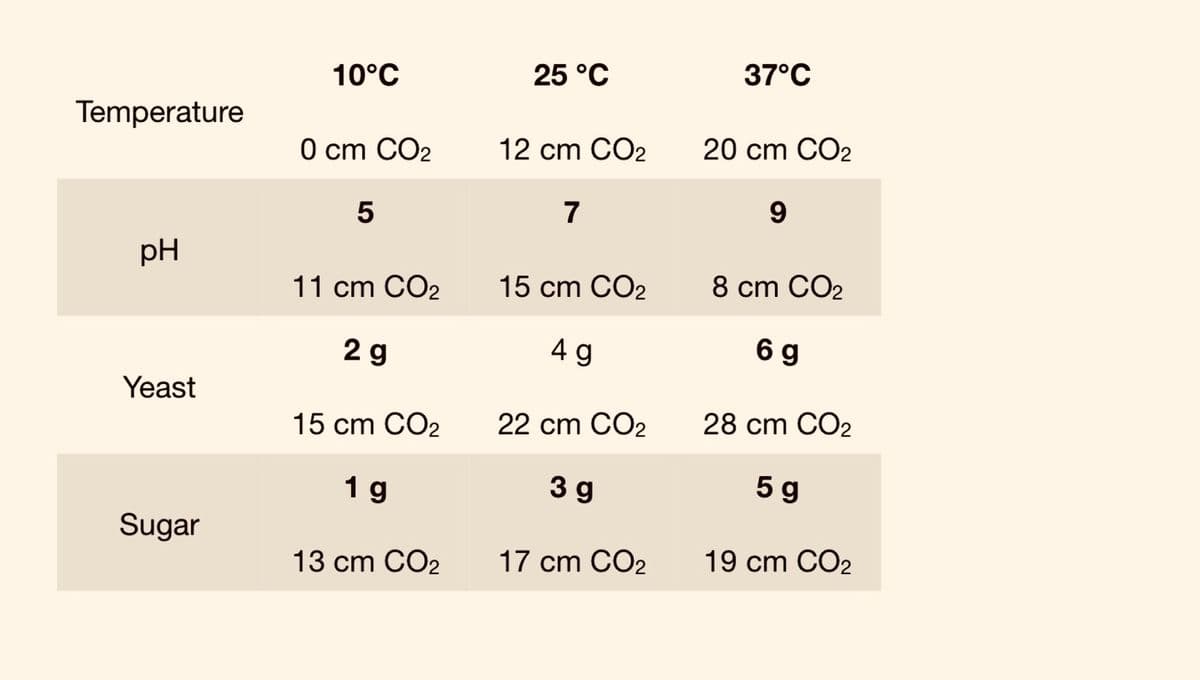
Q: ENZYME 1 ENZYME 2 Temperature Product Temperature Product Formation (C) (C) Formation (umol) (umol)…
A: An temperature increase increases the rate of reaction. An increase in temperature raises the…
Q: Phosphorylation of a molecule causes it to loose energy True False
A: It is a process of adding a phosphate group to a molecule. In living organisms, phosphorylation is…
Q: Which of the following molecules may be created through anabolic processes? O DNA O All of the above…
A: Metabolism is the chemical reaction occur in the organism's body to maintain their life. In an…
Q: the uploaded resources in the google drive for General Biology 1 or you may refer to the book…
A: Plants, algae, and some bacteria use photosynthesis to gather energy from the sun and convert it to…
Q: Compared to a solution of pH 9, a solution of pH 7 is Multiple Choice 2 times more acidic 20 times…
A: pH is a measure of acidity of a solution. A solution with high pH is more basic and a solution with…
Q: You conduct a redox reaction experiment with mutiple metals and nitrate solutions. One of the…
A: ANSWER;-
Q: 1.NAD+ is a nucleic acid, and NADH is a polysaccharide: True False
A: Introduction The most abundant carbohydrates in diet are polysaccharides, often known as…
Q: Which of the following reactions will occur only if coupled to a second, energetically favorable…
A: The occurrence in the cell allows one reaction to couple with another reaction, one of which is…
Q: What is the molarity of a solution if 360 g of glucose (C6H1206) is dissolved in 500 mL of solution?…
A: Molarity (M) is defined as the number of moles of solute per liter of solution. This is calculated…
Q: C&H12O6 (aq) → 2 C2H5OH(aq) + 2 CO2 (aq)+ 2 ATP (Glucose) (ethanol) (Carbon dioxide) (energy)
A: Fermentation is a process where sugars are converted to alcohol and carbon dioxide in anaerobic…
Q: please select and explain
A: In terms of temperature, available nutrients, acidity, salinity, and energy sources, Earth's…
Q: A fictional cubed-shaped bacterium, Bacterius cubis, occupies a volume of 2.8 femtoliters. This…
A: Given, A fictional cubed-shaped bacterium, Bacterius cubis, occupies a volume of 2.8 femtoliters.…
Q: H,N NH2 H,N NH, он H,N но OH
A: Protein is made up of different amino acids Joining. These amino acids are joined together by…
Q: organic compunds : 1. always contain oxygen 2. always contain nitrogen 3. always contain helium…
A: Answer is option 5.) always contain Carbon.
Q: 10 mL of 0.5 M CuSO₄ * 5H₂O solution from 10 M CuSO₄ * 5H₂O solution
A: CuSO₄ * 5H₂O solution is copper sulphate pentahydrate solution. The concentration of stock solution…
Q: Final Mass Initial Mass Difference Percent Difference 1 0.8 0.20 25.00 OM sucrose 0.1 M sucrose 0.9…
A: Diffusion is the movement of water from a region of low solute concentration to an area of high…
Q: Example of unfavorable enzymes reaction is the milk clotting? a. Ture b. False 2. Carbohydrates…
A: Microorganisms The submicroscopic mostly unicellular organism that are not visible by naked eyes…
Q: Binds here (1) and is changed to 2. Must bind here in order for reaction to occur Binds here (1) and…
A: Enzymes are essential biocatalyst present in living organisms. They catalyze different chemical…
Q: Which molecules in a plant contain nitrogen (select all) Glucose Protein Cellulose Nucleic acid
A: Nitrogen is one of major requirements of living beings but it can not be taken in the form in which…
Q: What wavelengths of electromagnetic radiation possess sufficient energy to be antimicrobicidal?…
A: Electromagnetic radiation travels as waves at maximum speed (speed of light). X-rays, gamma rays,…
Q: Label #1-5 with appropriate terms related to enzymes: Binds here (1) and is changed to 2 Must bind…
A: The enzymes are biocatalyst that perform specific reaction within the biological system. It is…
Q: Pure water will not freeze unit it reaches a temperature of about __________. Water in the liquid…
A: When liquid water is cooled, it contracts until a temperature of freezing point. Ordinarily, the…
Q: The basic type of reaction that forms all bio molecules is
A: Biomolecules or as they are known macromolecules in a cell are basically proteins, carbohydrates,…
Q: The total of ATP molecules that are produced during complete breakdown of two molecule glucose in a…
A: ATP Adenosine triphosphate, or ATP is a molecule that is involved in carrying energy between the…
Q: 4. Simple enzymes have inorganic cofactors, and conjugated enzymes have organic cofactors. 5.…
A: 4. Enzymes are bio-catalysts. They speed up a reaction by lowering the activation energy required by…
Q: Which of the following contain copper atoms (Cu2*) Select one: O a. Complex II O b. Complex III…
A: There are four enzyme complexes of ETC present in the inner mitochondrial membrane. Complex 1-…
Q: How many nucleotides are represented in the diagram? * 2 points 3 3 O 12 18
A: Nucleotides act as the building blocks of RNA and DNA. They also perform functions like metabolism,…
Q: I need help with my homework this question is very confusing How long would it take for a solution…
A: Concentration of carbohydrate in a solution is the moles of carbohydrate present in a specified…
Q: Choose the correct effect. * increase decrease not change Raising the temperature slightly will..…
A: Rate of a reaction: It is the speed at which the chemical reaction occurs. It is expressed as the…
Q: 0 1 5 6 7 6. 10 pH 1 2 3 The optional pH for enzyme 1 is: 2.
A: Optional pH :- The pH at which an enzymatic or process or any other reaction is most effective…
Q: 17. glycogen is a form of ____________in ________ cells long term- energy storage, plant…
A: Glycogen is the multibranched polysaccharides consist of multiple glucose molecules liked together…
Q: 1. A is usually based on either past experiences or information gained from reading peer-reviewed…
A: An experiment is a procedure used to demonstrate or prove the known existing fact, to prove the…
Q: Reactions 1: Y=0.2x + 0.8 Reactions 3: Y=0.2x + 0.5 1/Vmax = 0.8 1/Vmax = 0.5 Vmax= 1/0.8 mM/min…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation energy…
Q: CO2 (g) + H20 () CH1206 () + 02 (g) (a) How many grams of sugar are produced from 1000g (one liter)…
A: 6 CO2 + 6H2O → C6H12O6 + 6O2 This is the balanced equation of glucose synthesis using carbon dioxide…
Q: All proteins, 1 have quaternery structure made up of amino acids 3 are enzyme 4. made up of nucleic…
A: In nutritional biology, proteins represent one of the major macronutrients responsible for building…
Hi I need help creating a graph with these units. The graph should have labeled axis with units with a caption. Thank you.
Step by step
Solved in 2 steps with 4 images

- Which of the following temperature best suits for the growth and multiplication of transformed cells?a) 35°Cb) 36°Cc) 37°Cd) 30°CThis is what I did: to find change in concentration: 7.80e-18 - 4.50e-17 = -3.72e-17 Then applied diffusions law: (1.70e-9 x -3.72e-17 *5.20) / 1.00e-6 = -3.28848e-19 to convert to micrograms = -3.28848e-19 * 16g/mol * 1e6 mg/g = -5.26237e-12 micrograms.ODA stesa smal 3. What volume of a 2.5x10-³ M stock solution is required to produce 50.00 mL of a dilute solution that is 1.0x10-³ M? 10:noitos2/1otontant ds.I HIVA Experiment 8 emotiona zobas dgi inominoqx3 unit2000
- 13. 0.9% (m/v) NaCI solution and 5% (m/v) glucose solution are both isotonic to red blood cells. SHOW your work and watch sig figs & units. c. convert the concentration from M to % (m/v) for a 0.342 M NaC solution. (HINT: convert to g/ml and then multiple by 100%)Create one bar graph summarizing both the Tewameter and Corneometer readings (mention this graph in the text as Figure 1) Give a short description of Figure 1 as figure title (Figure 1) Label both axes On the graph, plot the average numbers and standard deviation, not the individual measurements Standard deviation should be displayed on the top of the average bars as an error barI do not understand the results of this experiment, explain what the results of this experiment are and the purpose Table 2 Absorbance vs CoCl2 concentration Test Tube Number Cobalt Chloride Concentration (mol/mL) Absorbance at 510 nm 1 0.000 0.000 2 0.009 0.024 3 0.018 0.055 4 0.027 0.085 5 6 0.036 0.045 0.112 0.138 7 (Unknown) 0.088
- Which of the phi/psi angles would be violating a ramachandran plot? Select all that apply: Phi= 90; Psi=-120 Phi= -120; Psi=120 Phi= 0; Psi=0 Phi= -90; Psi=-60 Phi= 120; Psi=-120choices for first blank (protonater,deprotonated) 2nd blank (higher,lower,same) 3rd blank (10,50,90,100) 4th blank (allowed from, excluded in)Expand the biological abbrevation (i) ACTH (ii) TSH.



