14 of 19 Review I Constants I Periodic Table limiting reactant. Part B How many moles of PCI5 can be produced from 25.0 g of P4 (and excess Cl2)? Express your answer to three significant figures and include the appropriate units. View Available Hint(s) HA ? 202 mol Previous Answers Submit XIncorrect; Try Again; 5 attempts remaining Your answer is the number of moles of P4. Now use this value and the coefficient for PCI5 in the balanced chemical equation to find the number of moles of PCl,. P Pearson F12 II F8 F11 F10 F9 F7 F6 & delete 7 } P U enter II K return ? shift +II
14 of 19 Review I Constants I Periodic Table limiting reactant. Part B How many moles of PCI5 can be produced from 25.0 g of P4 (and excess Cl2)? Express your answer to three significant figures and include the appropriate units. View Available Hint(s) HA ? 202 mol Previous Answers Submit XIncorrect; Try Again; 5 attempts remaining Your answer is the number of moles of P4. Now use this value and the coefficient for PCI5 in the balanced chemical equation to find the number of moles of PCl,. P Pearson F12 II F8 F11 F10 F9 F7 F6 & delete 7 } P U enter II K return ? shift +II
Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter11: Stoichiometry
Section11.2: Stoichiometric Calculations
Problem 11PP
Related questions
Question
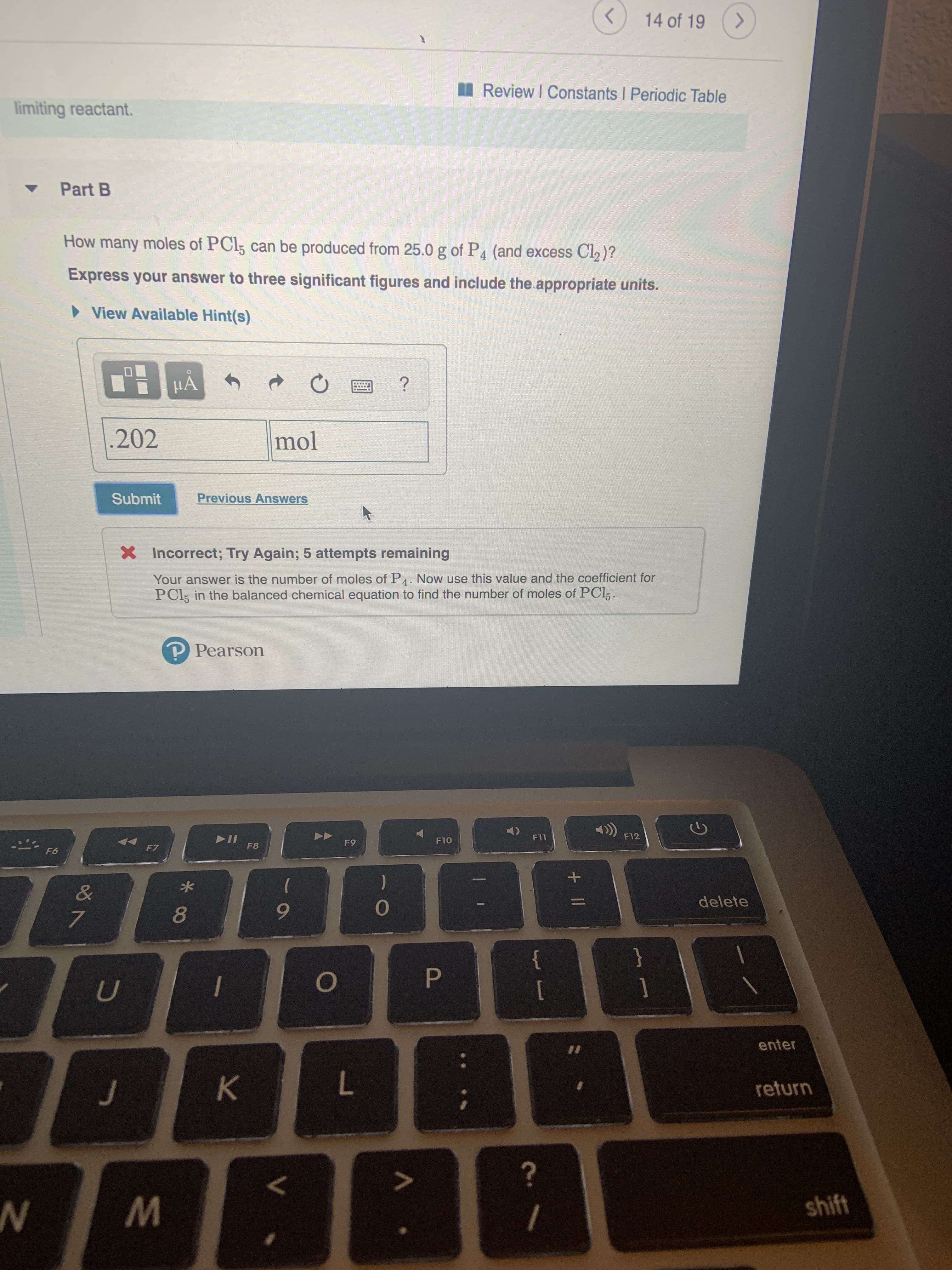
Transcribed Image Text:14 of 19
Review I Constants I Periodic Table
limiting reactant.
Part B
How many moles of PCI5 can be produced from 25.0 g of P4 (and excess Cl2)?
Express your answer to three significant figures and include the appropriate units.
View Available Hint(s)
HA
?
202
mol
Previous Answers
Submit
XIncorrect; Try Again; 5 attempts remaining
Your answer is the number of moles of P4. Now use this value and the coefficient for
PCI5 in the balanced chemical equation to find the number of moles of PCl,.
P Pearson
F12
II
F8
F11
F10
F9
F7
F6
&
delete
7
}
P
U
enter
II
K
return
?
shift
+II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning