19. If you used more than half of your NAOH solution in buret-refill It to the 0 mL mark on the fon If vou have more than 30 ml. Jeft. vou do pot need to refill the buret 1. Data Group1 Group 2 Group 3 Group 4 Group 5 Group 6 Volume of vinegar 2 25.00/25.0 25.00 / 25.00 sample 25.00/ 25.00 25.00/ 25.00 25.00/ 25.00 25.00/ 25.00 transferred, mL Molarity of sodium hydroxide solution, M 0.7784 0.7784/ 0.8161 / 0.8161 0.8211/ 0.8211 0.7784 / 0.7784 0.8161 / 0.8161 0.7784/ 0.7784 Final buret reading, 4 mL 26.90 / 26.73 24.10 / 25.65 23.85/ 24.00 28.10 / 27.85 26.20/ 27.00 29.10 / 28.80 Initial buret reading, mL 0.00/ 0.00 0.00/ 0.00 0.00 / 0.00 0.00/0.00 0.00/0.00 0.00/ 0.00
19. If you used more than half of your NAOH solution in buret-refill It to the 0 mL mark on the fon If vou have more than 30 ml. Jeft. vou do pot need to refill the buret 1. Data Group1 Group 2 Group 3 Group 4 Group 5 Group 6 Volume of vinegar 2 25.00/25.0 25.00 / 25.00 sample 25.00/ 25.00 25.00/ 25.00 25.00/ 25.00 25.00/ 25.00 transferred, mL Molarity of sodium hydroxide solution, M 0.7784 0.7784/ 0.8161 / 0.8161 0.8211/ 0.8211 0.7784 / 0.7784 0.8161 / 0.8161 0.7784/ 0.7784 Final buret reading, 4 mL 26.90 / 26.73 24.10 / 25.65 23.85/ 24.00 28.10 / 27.85 26.20/ 27.00 29.10 / 28.80 Initial buret reading, mL 0.00/ 0.00 0.00/ 0.00 0.00 / 0.00 0.00/0.00 0.00/0.00 0.00/ 0.00
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter11: Solutions
Section: Chapter Questions
Problem 25P
Related questions
Question
Chemistry HW need help. How to do these calculation base on the data that is given?
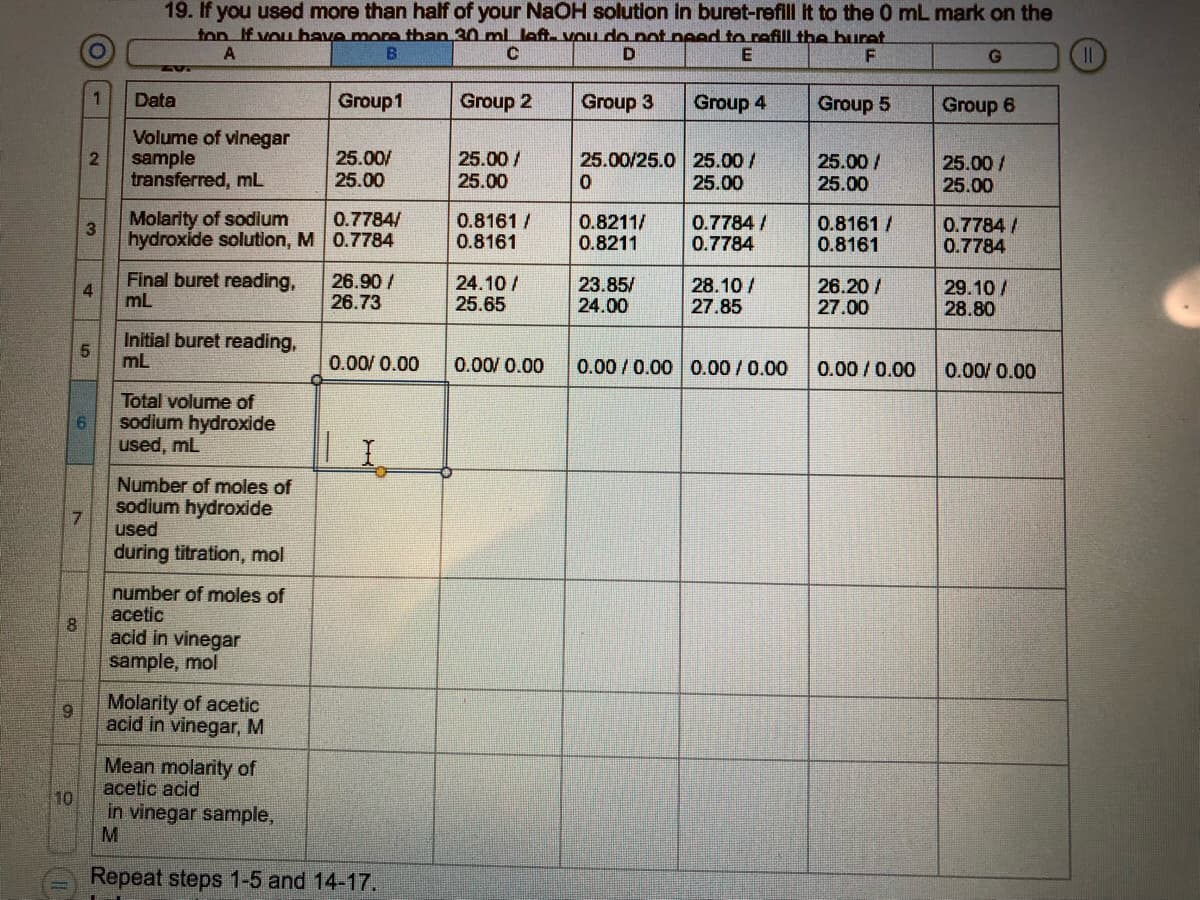
Transcribed Image Text:19. If you used more than half of your NaOH solution in buret-refill It to the 0 mL mark on the
ton If vou have more than 30 ml left- VOu do pot peed to refill the buret
A
B.
C
Data
Group1
Group 2
Group 3
Group 4
Group 5
Group 6
Volume of vinegar
sample
transferred, mL
25.00/
25.00
25.00/
25.00
25.00/25.0 25.00/
25.00
2
25.00/
25.00
25.00 /
25.00
Molarity of sodium
hydroxide solution, M 0.7784
0.7784/
0.8161 /
0.8161
0.8211/
0.8211
0.7784 /
0.7784
0.8161 /
0.8161
0.7784/
0.7784
Final buret reading,
4
mL
26.90 /
26.73
24.10 /
25.65
23.85/
24.00
28.10/
27.85
26.20 /
27.00
29.10 /
28.80
Initial buret reading,
51
mL
0.00/ 0.00
0.00/ 0.00
0.00/0.00 0.00/0.00
0.00/0.00
0.00/ 0.00
Total volume of
9.
sodium hydroxide
used, mL
Number of moles of
sodium hydroxide
used
during titration, mol
number of moles of
acetic
acid in vinegar
sample, mol
Molarity of acetic
9.
acid in vinegar, M
Mean molarity of
acetic acid
in vinegar sample,
10
Repeat steps 1-5 and 14-17.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
