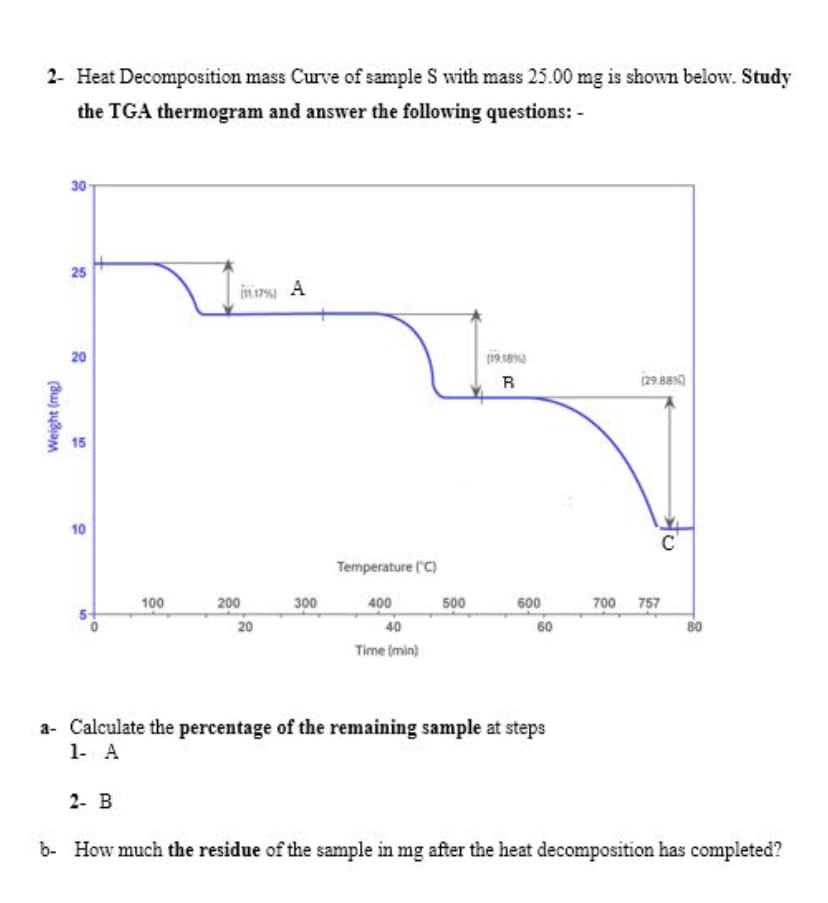
2- Heat Decomposition mass Curve of sample S with mass 25.00 mg is shown below. Study the TGA thermogram and answer the following questions: - 30 25 20 19 18 (29.880 15 10 Temperature (C) 100 200 300 400 500 600 700 757 20 40 60 80 Time (min)
Q: P*V (kPa*mL) pente (Ay/Ax) Volume (mL) 1/Pression (kPa^-1) Pression ( kPa ) 0.010343123 96.6826…
A: Percent error is the difference between the expected value and the actual value as compared to the…
Q: How many replicate measurements are needed to decrease the 95 and 99% confidence limits for the…
A:
Q: Sample Number of Fe content measurements mg/L on the sample 1 3 1.56, 1.45, 1.51 1.42, 1.39, 1.46…
A:
Q: 2- Heat Decomposition mass Curve of sample M with mass 3.335 mg is shown below. Study the TGA…
A: Given, The mass of the sample = 3.335 mg. The weight loss after A decomposition = 1.846 % The weight…
Q: Esall 0/50/ved its Volume (4L) His focusfooing Nagl from sodiumchloride it was related 3. by a daing…
A: Calculating the molarity of a diluted solution by using an equation given by dilution law: M1V1=M2V2…
Q: (2) j lubäi On the gravimetric determination of CI, if the weight of AgCl ppt. was 1.433 g.; the…
A: This method is used for the determination of the analyte based on its mass. The principle behind the…
Q: The oxygen mass transfer coefficient in fermentation vessels is determihed TPom measurements using…
A: Given, Cal' =0.25 mol.m^-3 with uncertainty 4%, Cal = 0.183 mol.m^-3 with uncertainty 4%…
Q: 2- Heat Decomposition mass Curve of sample S with mass 20.00 mg is shown below. Study the TGA…
A: Given, Mass of sample = 20.00 mg At each step the mass of the sample is getting deducted in a…
Q: 3.3. The specific gravity of gasoline is approximately 0.70 (a) Estimate the mass (kg) of 50.0…
A: (a)Specific gravity of gasoline, d = 0.7 g /ccVolume of gasoline, V = 50 L = 50000 mL
Q: 4) How would your calculated value of Kup be affected by errors introduced from the following…
A: Solubility product constant (equilibrium constant, Ksp) designates the attainment of equilibrium…
Q: a) State TWO (2) differences between those two calibration curves. b) State THREE (3) limitations…
A: A calibration curve or standard curve is a technique that finds out the strength of any substance…
Q: Study the tabulated data during an gravimetric analysis of BaSO4 experiment Trial T1 T2 T3 Mass of…
A:
Q: (a) During the analysis of water sample by argentometric titration, results obtained are as follows:…
A: Argentometric titrations are done using silver nitrate solution. It is usually done to determine…
Q: and
A: Given data set; Mass of empty flask = 351.74 g Mass of heated flask = 354.40 g Molar mass of acetone…
Q: The column was packed with 12 g of alumina. A 50:50 mixture of ferrocene and acetylferrocene was…
A: The experiment is about analyzing a mixture of ferrocene and acetylferrocene, quantitatively.
Q: Ordered: 1000 mL of 0.45% NaCl IV for 3 hours. Drop factor: 20 gtt/mL. Flow rate: ________ gtt/min
A: A question based on dimensional analysis that is to be accomplished.
Q: 2- Heat Decomposition mass Curve of sample S with mass 20.00 mg is shown below. Study the TGA…
A: A question based on TGA, which is to be accomplished.
Q: Amylum, commonly known as starch is a white powder with variable molecular formula due to the…
A: The number of moles of amylum, CO2, and water is determined as follows.…
Q: Soil sampled down to 1.8 m was mixed together then analyzed, and found to have 3.0 g C/kg on…
A:
Q: Using the Dumas method set-up, and eperiment was conducted using acetone ( MW=58.08). The mass of…
A: Dumas method is generally used to determine the molecular weight of volatile liquids. The method…
Q: Tirofiban (C22H36N2O5S MWt = 440.6) is present as tirofiban HCI monohydrate MWt=495.1) at 0.281…
A: Given the concentration of tirofiban HCl before infusion = 0.281 mg/mL Since we need tirofiban,…
Q: A solution composed of 50% ethanol (ETOH), 10% methanol (MEOH), and 40% water (H20) is fed at the…
A: Given: 50% ethanol,10 % methanol and 40 % water assuming no reaction in=outso starting with the…
Q: Observations: Burette Reading (ml) Volume of sample (ml) Normality of NazS203 solution (N) Volume of…
A: Given: Normality (N) of Na2S2O3 = 10.00 N Volume of the sample = 201 ml Volume (V) of Na2S2O3 = 4.5…
Q: One of the following is not one of the Minimization of errors. Internal standard method O الاختيارات…
A: Here we have to find which option is not correct I.e. one of them is not one of the minimization of…
Q: The homogeneity of the chloride level in a water sample from a lake was tested by analyzing portions…
A:
Q: 3. Gas Chromatography. Answer the following questions based on what you know about the GC in our…
A: The separation of the individual compounds in a mixture can be best accomplished by using the…
Q: The gravimetric factor used to express CoCBr6·H20 in a sample that is finally weighed as PbClBr is…
A: Gravimetric factor
Q: The column was packed with 12 g of alumina. A 50:50 mixture of ferrocene and acetylferrocene was…
A: Total mass of the mixture of (ferrocene + acetylferrocene) = 0.102 g
Q: Titration Heating Volume of DCIP (ml) Average Amt of Conc of #3 time vol of Vit C in Vit C Sample…
A: Ascorbic acid in Vitamin C reacts with DCIP in 1:1 fashion.
Q: C. Standardization Weight of vial and KHPH 59.0g Weight after 1st transfer 59-2 Weight after 2nd…
A: Molarity is defined as the moles of solute present in a liter of a solution. S.I unit of molarity is…
Q: Parameters Description or value Appearance of aqueous benzoic acid solution Concentration of aqueous…
A: Given that - Concentration of aqueous benzoic acid solution (g benzoic acid / 100 mL Solution) =…
Q: An 80 year old woman fell on the ice and fractured her femur. After several hours, when she arrived…
A: 1. Solution - According to the question - The pH is 7.31 which is more than neutral pH. So the…
Q: 25 um particles with a density of 2500 kg/m are suspended in a liquid with a density of 1000 kg/m at…
A:
Q: If 35,000 kg of whole milk containing 4% fat is to be separated in a 6-hour period into skim milk…
A: Given, Mass of whole milk = 35000 kg. % of fat = 4 Time period = 6 hours Skim milk fat = 0.46 %…
Q: the injector is kept at higher temperature than the column so that it will move quickly. If the…
A: A multiple choice question based on analytical separations that is to be accomplished.
Q: When trying to calculate the Kav for glutamate dehydrogenase I keep getting a negative value using…
A: Glutamate dehydrogenase is an enzyme which has a role in living beings to generate ammonia for urea…
Q: WATCH THIS TWO MINUTE VIDEO TO SOLVE…
A: A multiple choice question based on theoretical yield, which is to be accomplished.
Q: According to this, find the amount of CO2 absorbed by titration?
A:
Q: 5) SpectroPhoto meter device for meesuring Sample (2,4,6,81o pPm) at absapant cool,o.020-030.040.05)…
A: The absorbance vs concentration curve is drawn below.
Q: (5) If 200 g of polymer A, 300 g of polymer B, 500 g polymer C, and 100 g of polymer D are mixed,…
A: Molecular mass of polymer is a most important parameter and directly related to the physical…
Q: A series of calibration standards gave the following results. Peak Height (arb. units) % w/w H,0…
A: Gas chromatography is an analytical technique which is used to identify the analytes and their…
Q: Identify the effect of the condition on the indicated parameter. 1. A wet pipette was used to…
A: 1) A wet pipette was used to transfer 10.00 mL of 1M HNO3 in the determination of ΔHrxn. [magnitude…
Q: You have available a brown material (C:N ratio 60, 45% carbon, bulk density 0.3g/cm3) and a green…
A: Here, C:N ratio is directly proportional to the percentage of C Therefore, 60/25 = 45%/x Therefore,…
Q: b) The thermogravimetric curve for calcium salicylate monohydrate (CAC14H1006-H2O) is shown below.…
A: Thermogravimetry is an analytical technique that is based on mass lost from the sample when it is…
Q: . of T Determination. titriet acid and tedium tydriesxide. of the enthalpy of reaction Molority f…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: स 3. पG तnale ठेवतानe ० Pcतेवकাट प्न न एलकतिवबानकाट वते was melted व+ नि2 K, c4\c५ Rlc रमट Chamge in…
A:
Q: What is the literature value of the Ksp of Ca(OH)2 ? How far off from this value was your value(s)…
A: Ksp is defined as the solubility product and the general expression for solubility product is shown…
Q: Trials A Volume of Vinegar used (ml) 25.00 diluted to 250.00 mL Volume of Aliquot used (ml) 50.00…
A: CH3COOH + NaOH gives CH3COONa + H2O So, 1 mol NaOH neutralises 1 mol of acid.
Q: 2- Heat Decomposition mass Curve of sample S with mass 20.00 mg is shown below. Study the TGA…
A: TGA stands for thermogravimetric analysis or thermal gravimetric analysis. It is an analytical…
Q: Convert a speed of 973 cm/s to units of inches per minute. Also, show the unit analysis by dragging…
A: The conversion units required are 1S = 160 min 1cm = 12.54 in
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- A 100 g soil containing 20% smectitie mineral matter was mixed with 150 mL solution containing 0.10 M Pb. CEC of pure smectitie is 110 emol(+)/kg. CEC of the soil (cmol(+)/kg) is:The concentration of purified OXA-M290 is tested with a BCA assay. Serial dilutions of a bovine serum albumin (BSA) stock solution are prepared, then pipetted into a 96-well plate; each dilution of the BSA standard is tested in triplicate. Then, bicinchoninic acid and Cu2+ ions are added to all of the wells of the plate. After incubating the plate for 1 hour, a microplate reader is used to measure the absorbance of all of the wells in the plate at 560 nm. This generates the following data: BSA conc. (μg/mL), Replicate 1 Absorbance, Replicate 2 Absorbance, Replicate 3 Absorbance 40, 1.360, 1.403, 1.481 20, 0.750, 0.745, 0.810 10, 0.380, 0.344, 0.398 5, 0.198, 0.160, 0.183 2.5, 0.090, 0.100, 0.085 1.25, 0.038, 0.043, 0.051 0.625, 0.024, 0.028, 0.019 Prepare a calibration curve using these data. You can use Excel, R, SPSS or an equivalent graphing software. In this graph, plot absorbance (y-axis) against the concentration of the protein standard (x-axis). Calculate and plot…(a) Water in vadose zone contains chlorobenzene of concentration 40 mg/L. What is the equilibrium concentration soil air in contact with this solution? (b) An aquifer with porosity 0.3 has distribution coefficient of TCE as 100 L/kg. What fraction of TCE is in aqueous phase and sorbed phase of the soil?
- Various parameters in a gas absorption experiment are as follows; Water flow rate =5 L/min, Air flow rate=25 L/min, CO2 flow rate=6 L/min. Then, to find the amount of CO2 absorbed into the water 1 hour after the system is started and stabilized, 50 mL samples are taken from the inlet and outlet of the tank and titrated with 0.01M NaOH. 100mL samples are taken from the tower entrance and exit and sent to the hemp apparatus. The consumptions measured with the help of the Hemp apparatus are 2.1 mL (inlet) and 1.9 mL (output), respectively. According to this, find the amount of CO2 absorbed by titration? (Note: The relative error in the experiment is 10.)Explain and show work step by step: 1. A mixture of Al2O3(s) and CuO(s) weighing 18.371 mg was heated under H2(g) in a thermogravimetric experiment. On reaching a temperature of 1000 °C, the mass was l7A62 mg and the final products were Al2O3(s), Cu(s), and H20(g). Find the weight percent of Al2O3(s)in the original solid mixture.Amylum, commonly known as starch is a white powder with variable molecular formula due to the different number of repeating sugar units based on the plant that it is obtained from. A sample of amylum in the form of spherical beads was subjected to series of analyses to determine physicochemical properties in the quality control unit of a factory. From the results each bead is found to have a 4.5 mL volume and 1.47 g/mL density. 4 of the beads were placed in a reaction chamber combusted, as a result, 43.1 g CO2 and 14.7 g H2O were obtained. If the molecular weight of the amylum is 972.84 g/mol how many repeating units the starch molecule consist and what is the molecular formula of the starch? [Empirical Formula=CxHyOz, Repeating unit=n, Molecular Formula=( CxHyOz)*n= CnxHnyOnz]
- What could be the assumption when the MW of the sample does not match calculated molecualr weight from ∆Tb = mKb despite carefull data processing? The sample is not the compound of interest There are some impurities that affeced the boiling temperature Low heat is used to achieve boiling condition none of the assumption is valid 1 and III are valid assumptions all are valid assumptions I and II are valid assumptions10.5mL (mw 106) of benzaldehyde and 2.9g(3.63mL, mw 40) of acetone react with 5g of NaOH and 25mL of Ethanol to form 6.7g of Dibenzalacetone. What is the theoretical, actual and percent yield?Suppose you have a soil that is made up of 10% organic matter with CEC = 200 cmol/kg, 40% kaolinitewith CEC = 10 cmol/kg, and 50% vermiculite with CEC = 100 cmol/kg.1- Calculate the overall CEC of the soil by taking a weighted average of the three soil components.2- What percentage of the overall CEC is contributed by the organic matter? How does that numbercompare to the 10% of the soil itself that is organic?
- If to a solution of NaOH, in water and ethanol (20 ° C) 3 mmol of A (106.11 g / mol) and 2 mmol of B (58.06 g/ mol) and stirred magnetically for 10 minutes. After the reaction is completed, we proceed to isolate (work- up) and purify at C (246.29 g/ mol) yielding 0.300 g of the pure product. Calculate the% return for C showing his work. please show every single step and calculationsif a volumetric pipet delivers 24.844g of deionized water at 31.00 degrees C, what is the calibrated volume of the pipet?Downvoted for wrong solution. A river is carrying water containing 2000 mg/l Magnesium Chloride into a small lake. The lake has a naturally occurring Magnesium Chloride of 50 mg/l. If the river flow is 2500 Lmin and the lake flow rate is 1.5 m³.sec¹, what is the concentration of MgCl2 in the lake after the discharge point? Assume that the flows in the river and lake are completely mixed, that the salt is a conservative substance, and the system is at steady state.

