4) Write down the relationships your derived above as mathematical equations m = mass (g) AT = change in temperature (°C) q = amount of heat () 5) Use the relationships in question 4 to derive a proportionality statement containing all these variables. 6) Rewrite the proportionality statement has an equation that will have q on the left side and contain a constant "s" on the right side of the equation. The constant c is called specific heat.
4) Write down the relationships your derived above as mathematical equations m = mass (g) AT = change in temperature (°C) q = amount of heat () 5) Use the relationships in question 4 to derive a proportionality statement containing all these variables. 6) Rewrite the proportionality statement has an equation that will have q on the left side and contain a constant "s" on the right side of the equation. The constant c is called specific heat.
ChapterU5: Fire: Energy , Thermodynamics, And Oxidation-reduction
Section: Chapter Questions
Problem 12STP
Related questions
Question
Please help me complete question 4,5,and6.

Transcribed Image Text:4) Write down the relationships your derived above as mathematical equations
m = mass (g)
AT = change in temperature (°C)
q = amount of heat (1)
5) Use the relationships in question 4 to derive a proportionality statement containing all
these variables.
6) Rewrite the proportionality statement has an equation that will have q on the left side
and contain a constant "s" on the right side of the equation. The constant c is called
specific heat.
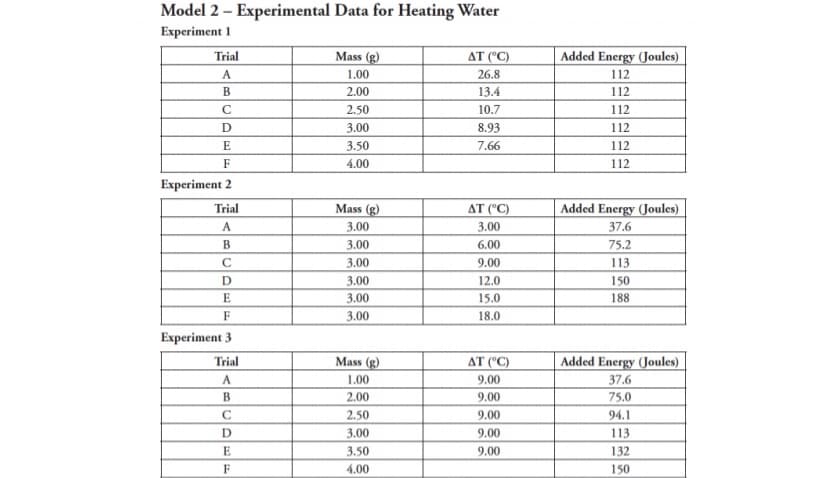
Transcribed Image Text:Model 2 – Experimental Data for Heating Water
Experiment 1
Trial
Mass (g)
AT ("C)
Added Energy (Joules)
A
1.00
26.8
112
B
2.00
13.4
112
2.50
10.7
112
D
3.00
8.93
112
E
3.50
7.66
112
F
4.00
112
Experiment 2
Trial
Mass (g)
AT ("C)
Added Energy (Joules)
A
3.00
3.00
37.6
B
3.00
6.00
75.2
C
3.00
9.00
113
D
3.00
12.0
150
3.00
15.0
188
F
3.00
18.0
Experiment 3
Trial
Mass (g)
AT ("C)
Added Energy (Joules)
A
1.00
9.00
37.6
В
2.00
9.00
75.0
2.50
9.00
94.1
3.00
9.00
113
E
3.50
9.00
132
F
4.00
150
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER
