A chemical engineer is studying the following reaction: HCH,CO,(aq)+CH,NH,(aq) → CH3CO,(aq)+CH3NH;(aq) At the temperature the engineer picks, the equilibrium constant K, for this reaction is 0.93. The engineer charges ("fills") four reaction vessels with acetic acid and methylamine, and lets the reaction begin. He then measures the composition of the mixture inside each vessel from time to time. His first set of measurements are shown in the table below. Predict the changes in the compositions the engineer should expect next time he measures the compositions. reaction compound concentration expected change in concentration vessel HCH,CO, 0.94 M t increase OI decrease (no change) CH,NH, 1.01 M t increase OI decrease O (no change) A CH,CO, 0.56 M O t increase OI decrease O (no change) CH,NH, 0.22 M O t increase OI decrease (no change) HCH,CO, 1.05 M t increase I decrease O (no change) CH;NH, 1.12 M O t increase OI decrease O (no change) CH,Co, O t increase 0.45 M I decrease (no change) CH.NH 0.11 M t increase OI decrease O (no change)
A chemical engineer is studying the following reaction: HCH,CO,(aq)+CH,NH,(aq) → CH3CO,(aq)+CH3NH;(aq) At the temperature the engineer picks, the equilibrium constant K, for this reaction is 0.93. The engineer charges ("fills") four reaction vessels with acetic acid and methylamine, and lets the reaction begin. He then measures the composition of the mixture inside each vessel from time to time. His first set of measurements are shown in the table below. Predict the changes in the compositions the engineer should expect next time he measures the compositions. reaction compound concentration expected change in concentration vessel HCH,CO, 0.94 M t increase OI decrease (no change) CH,NH, 1.01 M t increase OI decrease O (no change) A CH,CO, 0.56 M O t increase OI decrease O (no change) CH,NH, 0.22 M O t increase OI decrease (no change) HCH,CO, 1.05 M t increase I decrease O (no change) CH;NH, 1.12 M O t increase OI decrease O (no change) CH,Co, O t increase 0.45 M I decrease (no change) CH.NH 0.11 M t increase OI decrease O (no change)
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 12.41PAE: Because calcium carbonate is a sink for CO32- in a lake, the student in Exercise 12.39 decides to go...
Related questions
Question
100%
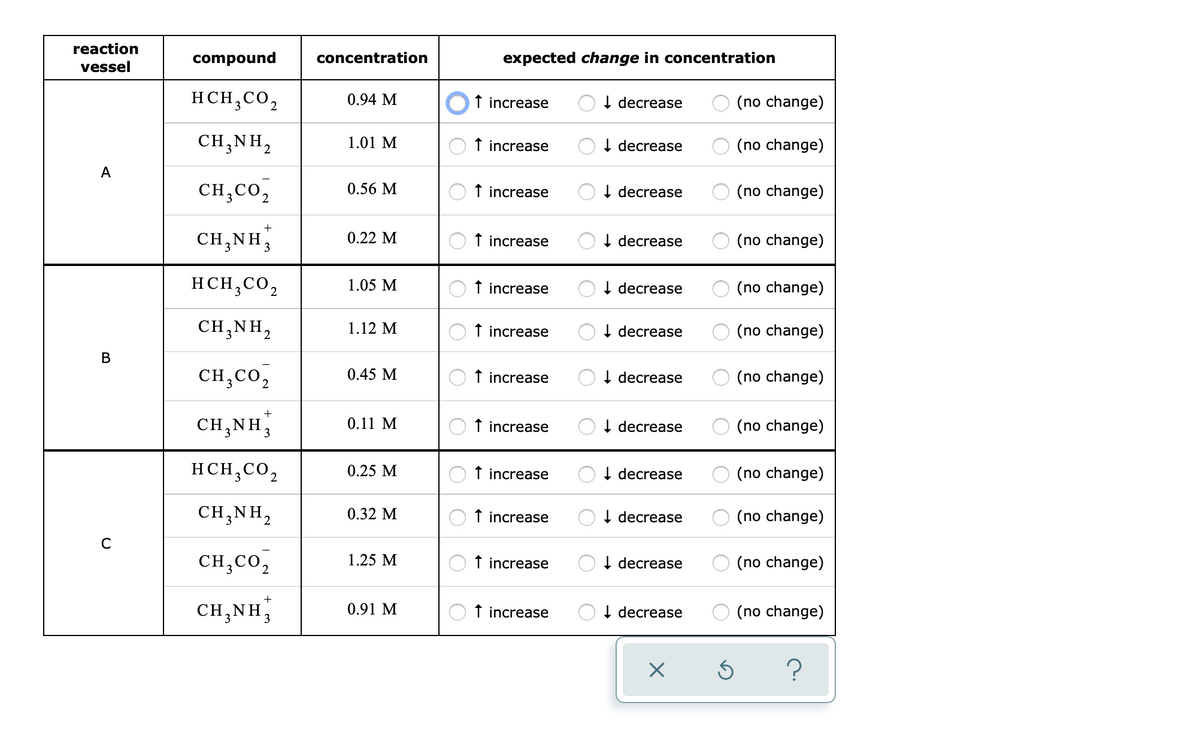
Transcribed Image Text:reaction
compound
concentration
expected change in concentration
vessel
HCH,CO,
0.94 M
f increase
I decrease
(no change)
CH,NH,
1.01 M
f increase
I decrease
(no change)
A
0.56 M
CH,CO,
f increase
I decrease
(no change)
+
CH;NH,
0.22 M
f increase
I decrease
(no change)
HCH,CO,
1.05 M
f increase
I decrease
(no change)
CH,NH,
1.12 M
↑ increase
I decrease
(no change)
CH,CO,
0.45 M
f increase
decrease
(no change)
+
CH,NH,
0.11 M
↑ increase
O I decrease
O (no change)
HCH,CO,
0.25 M
f increase
I decrease
(no change)
CH,NH,
0.32 M
f increase
I decrease
(no change)
C
CH,CO,
1.25 M
↑ increase
I decrease
(no change)
CH,NH,
0.91 M
↑ increase
O I decrease
(no change)
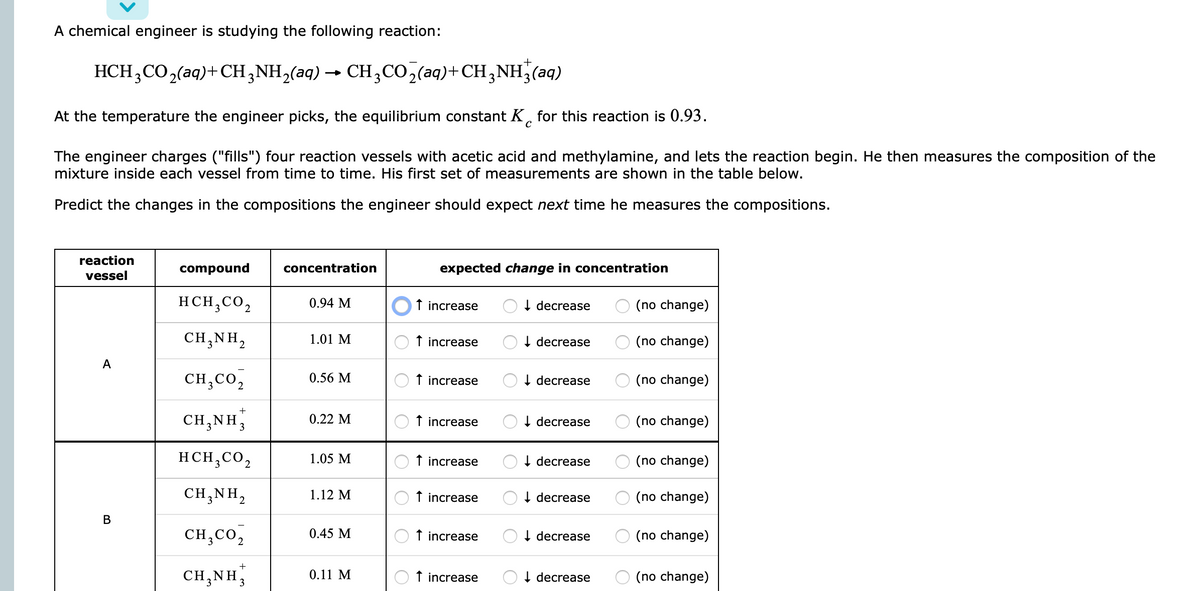
Transcribed Image Text:A chemical engineer is studying the following reaction:
+
HCH;CO2(aq)+CH,NH,(aq) → CH;CO,(aq)+CH;NH,(aq)
At the temperature the engineer picks, the equilibrium constant K, for this reaction is 0.93.
The engineer charges ("fills") four reaction vessels with acetic acid and methylamine, and lets the reaction begin. He then measures the composition of the
mixture inside each vessel from time to time. His first set of measurements are shown in the table below.
Predict the changes in the compositions the engineer should expect next time he measures the compositions.
reaction
compound
concentration
expected change in concentration
vessel
HCH,CO,
0.94 M
f increase
I decrease
(no change)
CH,NH,
1.01 M
f increase
I decrease
(no change)
A
CH,CO,
0.56 M
f increase
I decrease
(no change)
CH,NH,
0.22 M
f increase
OI decrease
O (no change)
HCH,CO,
1.05 M
f increase
I decrease
(no change)
CH,NH,
1.12 M
f increase
I decrease
(no change)
В
CH,CO,
0.45 M
f increase
I decrease
(no change)
CH,NH,
0.11 M
f increase
I decrease
(no change)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning