A key result from the kinetic theory of ideal gases is a calculation of the pressure of the gas in terms of the average kinetic energy of translation indicates the average value. As we will learn, particles in a gas have a range of velocities, so angle brackets indicate an average over the different velocities The result from kinetic theory expresses the pressure p in terms of the average kinetic energy of each particle, the number of particles , and the volume V of the gas. 2 N (KEtrans> Now for the Question: A room is filled with an ideal gas at a temperature T= 362 K and pressure p = 2 atmospheres (abbreviated atm). The dimensions of the room are 8 m x 8 m x 7 m. Note that 1 atm = 1.013 x 10° Pa, where a pascal (Pa) is a Nm (Newton, not number, per meter squared). Calculate the total translational energy, Utrans, of the N molecules in the room, where Utrans = N
A key result from the kinetic theory of ideal gases is a calculation of the pressure of the gas in terms of the average kinetic energy of translation indicates the average value. As we will learn, particles in a gas have a range of velocities, so angle brackets indicate an average over the different velocities The result from kinetic theory expresses the pressure p in terms of the average kinetic energy of each particle, the number of particles , and the volume V of the gas. 2 N (KEtrans> Now for the Question: A room is filled with an ideal gas at a temperature T= 362 K and pressure p = 2 atmospheres (abbreviated atm). The dimensions of the room are 8 m x 8 m x 7 m. Note that 1 atm = 1.013 x 10° Pa, where a pascal (Pa) is a Nm (Newton, not number, per meter squared). Calculate the total translational energy, Utrans, of the N molecules in the room, where Utrans = N
Physics for Scientists and Engineers: Foundations and Connections
1st Edition
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Katz, Debora M.
Chapter20: Kinetic Theory Of Gases
Section: Chapter Questions
Problem 72PQ
Related questions
Question
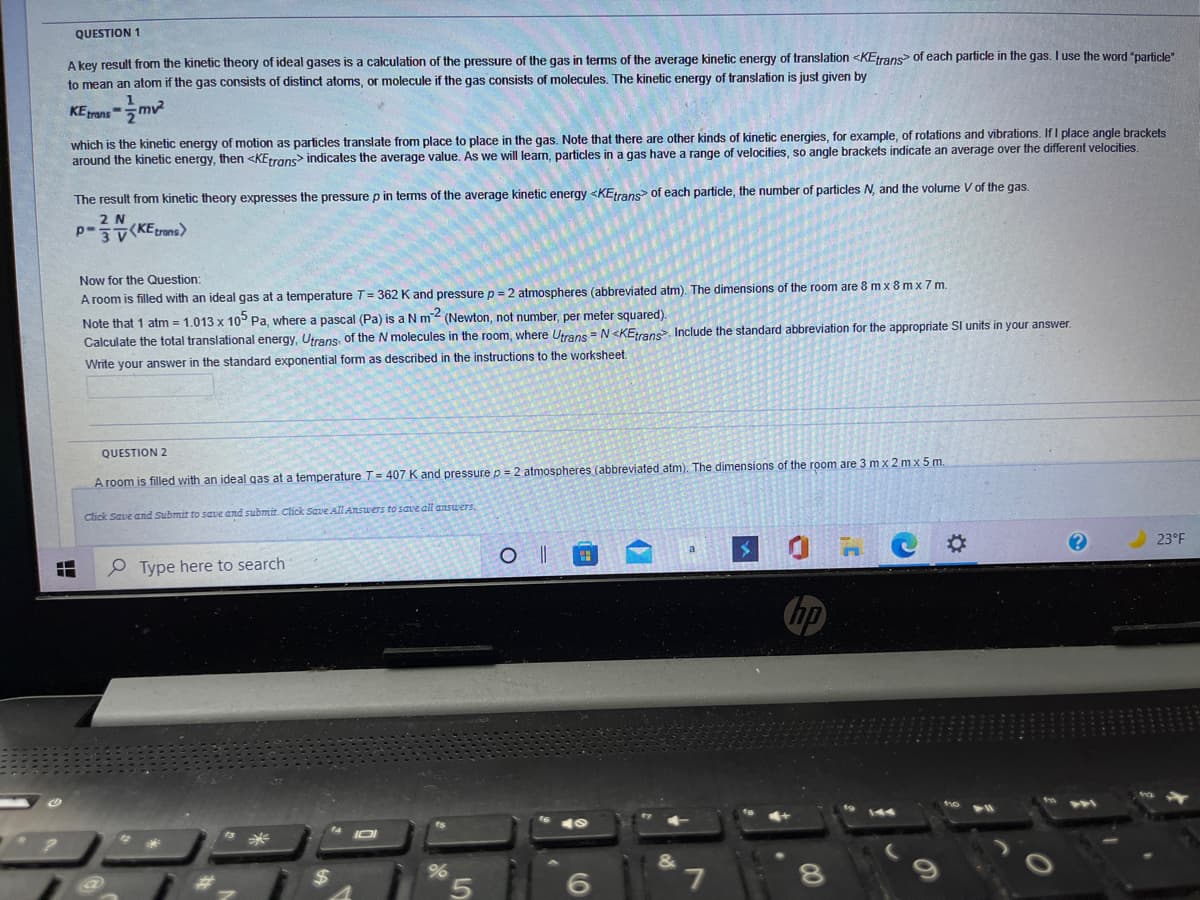
Transcribed Image Text:QUESTION 1
A key result from the kinetic theory of ideal gases is a calculation of the pressure of the gas in terms of the average kinetic energy of translation <KEtrans of each particle in the gas. I use the word "particle"
to mean an atom if the gas consists of distinct atoms, or molecule if the gas consists of molecules. The kinetic energy of translation is just given by
KEtrans
which is the kinetic energy of motion as particles translate from place to place in the gas. Note that there are other kinds of kinetic energies, for example, of rotations and vibrations. If I place angle brackets
around the kinetic energy, then <KEtrans> indicates the average value. As we will learn, particles in a gas have a range of velocities, so angle brackets indicate an average over the different velocities.
The result from kinetic theory expresses the pressure p in terms of the average kinetic energy <KEtrans of each particle, the number of particles N, and the volume V of the gas.
2 N
P-KErans>
Now for the Question:
A room is filled with an ideal gas at a temperature T= 362 K and pressure p = 2 atmospheres (abbreviated atm). The dimensions of the room are 8 m x 8 m x 7 m.
Note that 1 atm = 1.013 x 10° Pa, where a pascal (Pa) is aNm (Newton, not number, per meter squared).
Calculate the total translational energy, Utrans, of the N molecules in the room, where Utrans = N <KEtrans. Include the standard abbreviation for the appropriate SI units in your answer.
Write your answer in the standard exponential form as described in
instructions to the worksheet.
QUESTION 2
A room is filled with an ideal gas at a temperature T= 407 K and pressure p = 2 atmospheres (abbreviated atm). The dimensions of the room are 3 mx 2 mx 5 m.
Click Save and Submit to sae and submit. Click Save All ARswers to save all answers.
23°F
P Type here to search
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers with Modern …
Physics
ISBN:
9781337553292
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:
9781337553278
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College