
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
A sample in a 1cm cuvette gives an absorbance reading of 0.6. If the absorptivity for this sample is 15000 L/mol*cm, what is the molar concentration of the sample?
Expert Solution
arrow_forward
Step 1
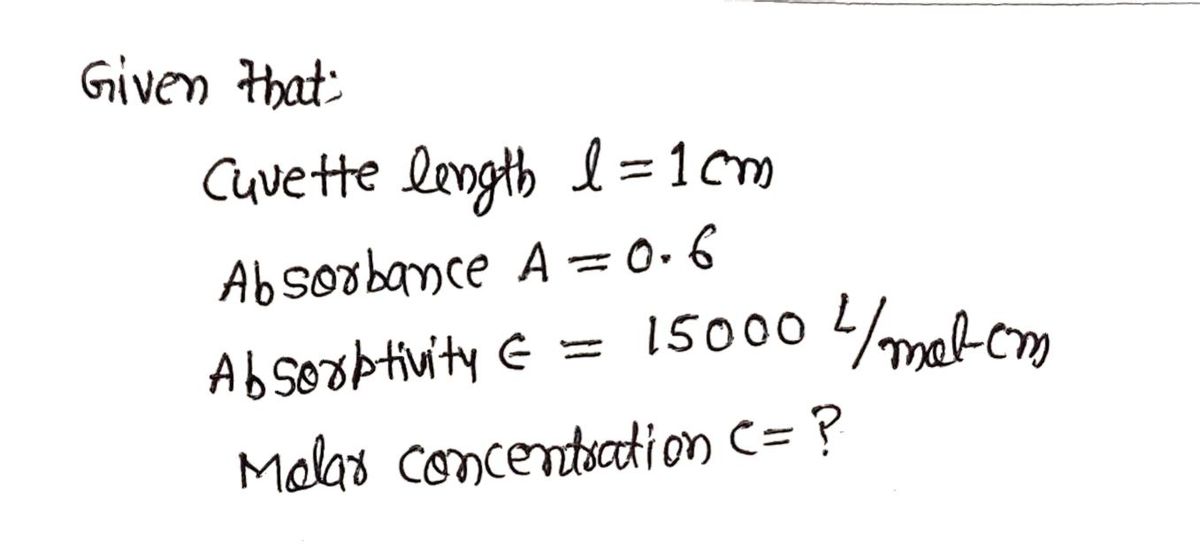
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Given this data, if a CV solution has an absorbance of 1.125, what is [CV]? [CV] / M A 1.0 × 10–6 M 0.250 3.0 × 10–6 M 0.750 Question 12 options: 4.5 × 10–6 M 5.0 × 10–6 M 5.5 × 10–6 Marrow_forwardA medical lab technician creates a standard solutions of percent hemoglobin in whole blood. The technician measures the absorbance of each solution at 520 nm and creates a standard curve of the absorbance versus percent hemoglobin in whole blood and finds the linear equation below, with an R2 of 0.9985. y=0.0523x+0.072 A patient sample has an absorbance of 0.915. Using the standard data curve data, calculate the percent hemoglobin in the samplearrow_forwardA 3.7 × 10−4 M solution of compound X has an absorbance of 0.5342 when measured at 740 nm. Assuming a pathlength of 1 cm, calculate compound X’s molar absorptivity.arrow_forward
- * Question Completion Status: QUESTION 6 Most analytical machines used in science can either measure absorbance of light, like the spectrophotometers used in BioZ 151 and 152 labs, or can measure change in electrical conductance. In order to convert things like absorbance values into concentration data, a linear regression is used. In order to determine nitrite concentration in water samples, nitrite is reacted with several chemicals to produce a purple color. The absorbance of the solution is measured from known amounts of nitrite to produce a standard curve to analyze samples. Use the data in the provided table, or from the linked spreadsheet to make a graph and perform a linear regression using treadline. mg/L NO, Absorbance 0.1 0.183 0.3 0.5 0.7 0.542 0.914 1.298 1.61 In order for this to work, samples must be diluted prior to measurement standard curve. If a sample underwent a 0.02 dilution, and produce an absorbance value of 0.903, what is the resultant concentration of NO2 in mg…arrow_forwardIn polarimetry, indicate the correct explanation of the drawing. (1). No light reaches the eye, since the analyzer prevents it. (2). The maximum angle that the plane of polarized light can be deflected by any species is ±45°, since 45° + 45° = 90° and sin 90° = 1. 00/07 (3). The polarimetric tube has a levorotatory substance. (4). Any molecule near the face through which the polarized light enters deviates its plane less than any molecule that is close to the opposite face, due to the longest optical path traveled by the light. Oarrow_forwardTonic water (20.0 mL) was added into each of two volumetric flasks (100 mL). The first flask was made up to volume with deionised water and produced an absorbance of 0.200. To the second flask was added an aliquot (20.0 mL) of a quinine standard solution (20.0 ppm) and after being made up to volume this produced an absorbance of 0.400. What is the concentration of quinine in the tonic water? a. 8.00 ppm b. 800 ppm С. 40.0 ppm d. 400 ppm е. 20.0 ppmarrow_forward
- em1.png Part 3: Q9 Average absorbance Flask 8 Flask 9 Flask 10 1:1 Ratio so, Flask 11 Part 3: Q Flask 8 Flask 9 Flask 10 Flask 11 Part 3: Q10 Flask 8 Flask 9 Flask 10 Flask 11 Part 3 0.146 0.166 [Fe] -0.002mol/L 0.215 0.294 [FESCN³mol/L) 4.59% 10 5.18*10* 6.64*10* 9.08*10* [FXmol/L) 5.00 *10* 5.00*10* Y=3362x-0.0083 R² = 0.9993 5.00*10* 5.00*10* (Amounts of flask 8 (9 - 11's values are in the spreadsheet) 71% volume 5.00mL Molarity of Fe(NO3))3 = 0.2M Volume of Fe(NO3))3 = 5.00mL Moles of Fe³= Molarity x volume(L) Moles of Fe¹ = 0.2M X 0.005L = 1x10-³mole x [SCN][mol/ 4x10 5×10-4 7x10 1×10 1 Concentration of Fe³ = mole of Fe³ Volume mole of Fe³1x10³ mole G.O05L 8, Average absorbance for solution 8 (same for 9 - 11) Average absorbance for flask 8 = sum of all absorbances / number of readings (0.145 +0.147 +0.148) 3 Using equation of calibration curve's line(slope) of best fit, the average absorbances of the unknown concentrations to determine [FeSCN-Jat equilibrium in solution 8:…arrow_forwardHow do I find the theoretical molarity?arrow_forwardGiven: You mix together 3 mL of Al3+ (3.00 x 10-5 M) and 3 mL of xylenol orange (5.00 x 10-5 M). The absorbance at room temperature is 0.1 and the absorbance at 65oC is 0.35. a. What is the concentration of AlQ- at 65 degrees Celcius?arrow_forward
- Chemistryarrow_forwardA standard containing 20 mg/mL of compound P is placed in a 1 cm cuvette. The absorbance of the standard is 1.20 at 600 nm. A sample containing Compound P has an absorbance of 0.50. What is the concentration of this sample?arrow_forwardSample #1 has an absorbance of 0.300. Sample #2 is the same substance and has an absorbance of 0.600 using the same equipment. What is the relative concentration of the second sample? Select one: a. The concentration of sample #2 is the square root of that of #1. b. The concentration of sample #2 is that of sample #1 squared. c. The concentration of sample #2 is half that of sample #1. d. The concentration of sample #2 is twice that of sample #1.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY