Q: concentration of 1.8 kg/m3, with the concentration of copper in the lixiviant that is irrigated onto…
A: Given: Average grade is 0.7% Total pregnant solution is 8000 m3/hr Average concentration can be 1.8…
Q: dm3 Reaction rate specification ,K ( mol. min FA0 – FAI = -rAV kCA? = -TA voCAO – v0 CAI = kCA1?V vo…
A: We have the second-order reaction 2A→Product Rate expression for this reaction is -rA=KCA2 Mass…
Q: II Rb Courses- Blackboard Learn > General Psychology - Fall 2021 A ALEKS - Griffin Barden - Learn…
A: Given that : The heat energy absorbed by the chemical reaction = 350 kJ The heat energy flowed in…
Q: Kw = K,K, %3D Example: The HPO,2 ion has a Ka of 1.3 x 10-3 at SATP. What is Kb for the PO,3 ion?
A: Kw= Ka × Kb Kw = water dissociation constant Ka= Acid dissociation constant Kb= Base dissociation…
Q: 共 aterally vatne t o side F 31 2-Gmaller the osu ius 39-kemder gasho Cnemus by hoae ue mbe. -#37…
A:
Q: There is actually something im confused about, when you calculating V0 for S0=200uM --> V =…
A: Actually, that is a typo that needs correction. Since we are calculating the initial reaction rate,…
Q: Quanktahre Amalyais : Ex: A constant eectic cunent depasite 365 mg. 9 Ag in 216 mimutes yom an…
A: Given that: amount deposited= 365 mg t = 216 min To find: the amount of current?
Q: O D. rate = k[X2] O F. rate = k[Aa[X- %3D
A: Rate law is experimental calculated expression which contains concentration of reactants raised to…
Q: If the instability constant of EDTA is 8.35 x10-22, the stability constant will be ---- O 8.35 x1022…
A:
Q: BA 125 ml. tank in charged with owygen until the prei 730t 2C. Cile en in the tank 319 051
A: A gas can be described based on 4 parameters, that is pressure, temperature, volume and number of…
Q: Given the following balanced reactions: P205 + 9H20 → 2PO43 + 6H30* 2PO43- + 6Ag* → 2Ag3PO4(s) Ag* +…
A: Balancing a chemical equation is necessary so as to abide by the law of conservation of mass and to…
Q: If an intravenous infusion contained 30000 units of heparin sodium in 1000 mL of D5W and the rate of…
A:
Q: hat the time ad submevs ion in day of Brass lloy(25%2n, 75%. cu) , avca was 10 cm², that 2. 5 kg had…
A:
Q: NazCOs(OHf – \130,9) (AG^F -1047.7)(S°136.0) BINA2CO3IS) + 21HNO3(ag) →2 Na N03(aq)+COsgitHz0ce)…
A: For the thermodynamic reaction ∆G = ∆H-T∆SFor the calculation of ∆Go =…
Q: क Rb ALEKS Chemistry- 2021FA-CHI X A ALEKS - Griffin Barden - Learn…
A:
Q: A ceric sulfate solution that was standardized using 200 mg of arsenic trioxide (MW 197.84g/n)…
A:
Q: 1. The Ksp of Ca3(PO4)2 is 1.3 × 10−26. Estimate the solubility of this salt in units of g. L−1…
A:
Q: Which of the following are equivalent to 2,500 ppm Cu2+? (There may be more than one answer)…
A: Given data 2500 ppm equal to
Q: Examples Example (1);- The following data obtained from decomposit of H2 02 in aquees Solution:…
A: Please find the graph attached here with the solution .
Q: T, K k, s¹ 273.15 5.6 x 10-6 283.15 3.2 x 10-5 293.15 1.6 x 10-4 303.15 7.6 x 10-4 Using linear…
A: Arrhenius Equation: LnK= LnA - EaRT Comparing with, Y= mx + c Y axis= K X axis= 1T
Q: 17- The percentage of profein in meat products is determined by multiplying the %N as determined by…
A: Given, weight of meat scrap = 2.000 g Vol of H2SO4=50.0 mL Vol. of NaOH = 28.80 mL
Q: (a) the only information about the precision of the method is the precision for the three data.…
A: Mean x¯=7.23+6.95+7.533=21.713 =7.236 or 7.23 Standard deviation (S)=∑(xi-x¯)2n-1…
Q: Styles Part I: Separation of Ammonium Chloride Mass of small evaporating dish Mass of small…
A:
Q: A group of students compiled the data shown in data table 1 below. What is the exact calculated…
A: Order with respect to IO3-(aq) can be calculated using rate law equations for experimental data 1…
Q: Nyroysis reoction is given bela : PPP ty pp toP a Now long will it take for D01% qf pp to hyarolize-…
A: The given hydrolysis reaction is a consecutive reaction in which initial concentration of substrate,…
Q: How do you find the delta Hrxn in J/mol?
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: The oxidatiun of iodide ion by hydrogen peroxrde by in an acidic soution is described the balanced…
A: H2O2(aq) + 3I-(aq) ----> I3-(aq) + 2H2O(l) rate = k [H2O2]x [I-]y
Q: CuCl2 + AgNO3 → + No new data to save. Last checked at 4:58pm. Submi
A:
Q: E MasteringChemistry: HW 01b C Get Homework Help With Che b My Questions | bartleby > MyLab and…
A: Density of a substance is the ratio of mass of the substance to the space occupied by it. Using…
Q: Multi ple choce queaiors 12. 10C Of y Chaige passes a point na Crcu in 2 mamtea he current Ip the…
A: Given, Amount of charge = Q = 10 C Time = t = 2 minutes Current in the circuit = I = ?
Q: Some wastewater stream contains 0.015 M Hg22+. Some chemical engineer decides to remove it by…
A: Concentration of Hg22+ in waste water = 0.015 M Volume of waste water taken = 100 mL Concentration…
Q: A ceric sulfate solution that was standardized using 200 mg of arsenic trioxide (MW = 197.84g/n)…
A:
Q: c. MW 88, C4HeO2 48 20 4800 35ee 250e 2000 1502 1200 OSTC 171
A:
Q: dt The Gompertz growth model is commonly used to model tumor cell growth. Let v(t) be the tumor's…
A:
Q: What is the 95% confidence interval for the mean of the data, assuming
A: (χi - μ) (χi - μ)2 1 3.55 0.10 0.010678 2 3.65 0.20 0.041344 3 3.14 -0.31 0.094044 Mean…
Q: 3. After correction for water conductivity, the conductivity of a 2.50 x 104 M aqueous solution of…
A: The reaction taking place is given as, => Mg2+ (aq) + SO42- (aq) -------> MgSO4 (aq) Given:…
Q: Consider the calcination of CaCO;: CaCO3 -> CaQ + CO2 Before calcination, the initial weight of…
A: The reaction is process by the reaction between two or more reactant to each other. The mass of each…
Q: Ksp=4.1x10-36 for Pb3(AsO4)2 Calculate Eo for Pb3(AsO4)2(s) + 6e- 3Pb(s) + 2AsO4 3- Pb2+ +2e- …
A: Given data: KSP =4.1x10-36 E0pb2+/pb = -0.126V Number of electrons = 6 e-
Q: nformation Securit X A ALEKS - Ciapha Dorley - Learn…
A: The rate constant (k) for the reaction is calculated as shown below where r is the initial rate of…
Q: Virtual Exp: Hess' Law Report Page: Table 1: Measurements foom aup- Reaction mass NaOH Tinitial (°C)…
A:
Q: A soil is analyzed for total iron content by dissolving 0.1 g of soil in 10 mL of acid and digesting…
A: Amount of soil = 0.1 g Final volume of solution to be analyzed = 100 ml Mass of iron present in 1000…
Q: aplete this table relating the values of Ecell and AG to the Q/K ratio. QIK Ecll AG 0 > 1
A:
Q: Chrome File Edit View History Bookmarks Profiles Tab Window Help Mon Oct 25 6:23 PM * Groups - HSCI…
A:
Q: OneLogin ALEKS - Kayley Valentine b nickel (II) sulfate cation and ani x Lea x + A…
A: A chemical compound consists of two or more different elements which are bonded with each other…
Q: 75A empan is considering purchasing a new machine for Sos0 000 hat will increase the firm s ncome by…
A: Soln
Q: If the amount of a chemical in a lake (4 tonnes) and the inflow (7) Kg/yr, what is the residence…
A: Given: The amount of chemical in the lake, (M)=4 tonnes=4000 kg. The flow rate, (F)=7kg/yr.
Q: Anna analyzed arsenic content on a fish purchased from local wet market. According to Food…
A: Arsenic in water often occurs as a contaminant in fishes.
Q: ALEKS ← → C x A ALEKS - Tanvi Damle - Learn X 505 Missing Women.pdf X b Answered: A mixture of…
A:
Q: Complete the following operations by filling in the exponent for the result: (*)(-*)--O %3D = k y…
A:
Calculate the cell potential of each of the following
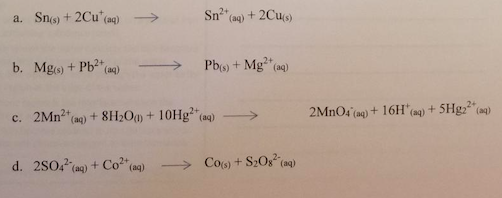
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- In a river with a constant flow rate, the factory releases wastewater containing NaCl at a concentration of 200 mmol / L to the river at 20.0 L / s. The Na+ and Cl- concentrations at the downstream observation point were 1.00 mmol / L and 0.800 mmol / L, respectively. What is the river flow Q and Cl- concentration before the inflow of drainage? The river originally contains 0.500 mmol / L of Na+.Beaker 0.00200 M Fe(NO3)3, mL 0.00200 M NaSCN, mL total volume, mL 1 3.000 2.000 10.00 2 3.000 3.000 10.00 3 3.000 4.000 10.00 4 3.000 5.000 10.00 5 (blank) 3.000 0.000 10.00 In Solutions 1-4 you are adding successively larger volumes of 0.00200M SCN- to the Fe3+ solution and diluting to 10.00 ml. Calculate the final diluted molarity of SCN- in solution #1 Your answer should have 3 sig figs =150 kmol of an aqueous phosphoric acid solution contains S mol percent H3P04. The solution is concentrated by adding pure phosphoric acid at a rate of 20L per minute. Write a differential mole balance on phosphoric acid and provide an initial condition. Solve the balance to abtain an expression vdor np(t) . Use the result to derive an expression far xp(t), the mole fraction of phosphoric acid in the solution. How long will it take to concentrate the solution to IS percent H3P04 ?
- Use the following atomic masses (in g/mol): K = 39.1; Mn = 54.94; O = 16; H = 1; P = 30.97; C = 12.01; Na = 23; Mg = 24.31; S = 32.06; Zn = 65.38; Cu = 63.55 In the standardization of Hydrochloric Acid against pure anhydrous Sodium Carbonate, 1mL was found to be equivalent to 0.05g of the primary standard. Compute for the normality of the acid.There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…
- Electrolytic manganese dioxide can be prepared from manganese carbonate ore by crushing,milling and leaching the ore in sulphuric acid. Manganese sulphate is crystallised from thesolution, redissolved and electrolysed to give the manganese dioxide.If the crystallisation were performed in a 30m3 tank and the concentration of the solutionentering the tank were 160 grams per litre and left the tank at 40 grams per litre, how muchMnSO4.5H2O would be produced.Stock iron(II) solution (200Ug mL-1 Fe) ferrous ammonium sulfate hexahydrate mass= 0.1437g, transfer it to a 100 ml beaker. add 15 ml approx of water and 15m1 'approx of dilute sulphuric acid (2M H2SO.). then transfer FeII to 100 ml flask makeup to the mark with water. calculate the moles of ferrous ammonium sulfate hexahydrate solution in unit ug/mL.



