Q: C15T05Q5196 Give the minor product(s) of the following reaction. CH3CH2CH2CH2CI (1 mole) AICI3, heat
A: In alkylation of isopropyl benzene, lewis acid generates carbocation by forming a lewis acid base…
Q: Find the molecular weight of the following compounds **. You have to do the complete procedure as…
A: We know that Molecular weight : molecular weight is the sum of mass of all the atom in a molecule.…
Q: Answer 6, 7 and 13
A: 6--- NH3 7----Ammonia 13---Sn(OH)4
Q: 4. ОН SOCI, pyridine ?
A: SOCl2 in pyridine gives SN2 mechanism with alcohol..in this reaction we got inverted product...
Q: -3.46 What are the molecular masses of the following pharmaceuticals? (a) C33H35FN2O5 (atorvastatin,…
A: Data : The mass number of C= 12 The mass number of O=16 The mass number of F=19 The mass number of…
Q: Rank the following in o H.
A: Carboxylic acid : R-COOH is a carboxylic acid functional group Acid derivatives: 1. Acid halides…
Q: The figure below shows TEM micrograph of a rubber toughened polystyrene (PS) orthotic (i.e. rubber…
A: Diffrential scanning calorimetry: Differential scanning calorimetry is a technique in which the…
Q: Why is the ΔHvap higher than ΔHfus for any given compound?
A: To explain : Why is the ΔHvap higher than ΔHfus for any given compound?
Q: Acetylene burns in air according to the following equation: ΔΗ. AH СН (g) + O23) — 2 СО,(g) На0(g)…
A: The standard free energy of formation refers to the energy change that occurs when a compound is…
Q: nformation to answer the following questions. ydrocarbon AHcombustion (kJ/mol) -890 -1560 -2219…
A: Balance Chemical equation means no of atoms should be equaal in both side reactant and product…
Q: 23
A:
Q: Pls
A: Equillibrium constant is the ratio of concentaration of products to the concentration of reactants.
Q: How des hy drgan bonding afecd plynerizing ? Vfolymer
A: Hydrogen bonding is the bonding between Hydrogen and electronegative atom that is Fluorine, Oxygen,…
Q: Given the following: = Sa (0) + O2(g) → 50₂ (0) 3 (a)+02 (0) 501 (9) The heat of reaction for SO₂(g)…
A: The enthalpy change is a state function and the Hess law states that the change of enthalpy in a…
Q: ction sequence shown belo (1) Br2, FeBr3 (2) Mg/ether QH он (A) (B) (3) (4) dil. H* /H,O (C) (D) он…
A: Ans: compounds A and B
Q: B-Carotene is better to be absorbed through cooked food? A. True B. False
A: We can control the amount lost by using specific methods of cooking. Beta carotene is a fat-soluble…
Q: Answer Q35, 36, 37
A: Answer 35: Dilatometry is the study of the volume changes occuring during the chemical reaction.…
Q: 5. If a student obtained 2.45 but expected 2.66, what is the percent yield? 6. What three functional…
A:
Q: ??? d) d) :OH
A: this is the nucleophilic addition reaction
Q: enthaipy of fomation of pneno enthaipy formation of pienoi gaseous Carb -393.51 kJ mol1 2C6H5OH +…
A: Given-> 2C6H5OH +15O2 -----> 12CO2 + 6H2O I think it asked standard enthalpy of given…
Q: The vinyl chloride molecule is (C2H3CI)n Calculate the molecular weight of polyvinyl chloride (PVC)…
A: The sum of atomic weights of all the atoms of the molecule is its molecular weight.
Q: OH OH and and and and OH L
A: In functional isomerism, the molecular formula should be the same but change in the functional…
Q: Q12- At pH=7.89 A) [H₂L]=[L²] B) [HL]=[H₂L] C) [H₂L]> [HL] D) [L²] = [HL]
A:
Q: 30. OO The above is an example of a(n) a. aliphatic compound O b. triphenyl compound c. haloaromatic…
A: The given compound contains benzene rings, so it is an aromatic compound. The number of benzene…
Q: Answer Q43, 44, 45
A: The correct answer of given question are given in step 2
Q: Answer Q48, 49, 50
A: 48. Alcohols reacts vigorously with PCl5, secondary alcohol when react with K2Cr2O7 gives Z is a…
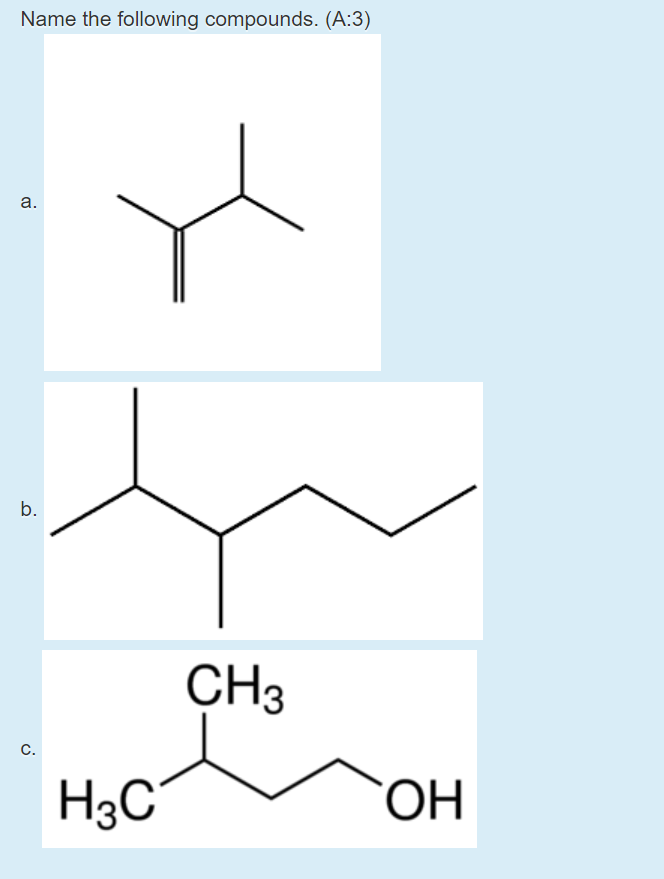
Q: Name the following compounds: (a)(b)(c)
A: Given: structure of molecules
Q: Consider the following equation that is commonily used in pharmacokinetic studies of drugs In[D]e =…
A: To solve this problem first we have to rearrange the given equation to get K
Q: Methorimeprazine CNozinan) an antipsy choie drug has beern 0rdered fur your Cclient. The order reads…
A: A numerical problem in solution, which is to be accomplished.
Q: I need explanation with hand written answer pl
A:
Q: BMy Dashboard myE X Bb Unit 9 - Molecular GeX G charge on Nh4 - Goo X Pt Dynamic Periodic Ta x New…
A: Since there are multiple sub-parts, we are authorized to answer only the first three sub-parts. If…
Q: LOD TRANSHETTANCETAT D 4000 3000 N Compound 1 2000 Compound 2 HAVENUMBERI- 1500 OH Compound 3 imppan…
A: Given : ir spectrum And structure of compounds
Q: h of the following chemicals have sp carbon? t one or more: N. H;C CH3
A: sp is a type of hybridization.
Q: In the enperiment on nile sed, would flousoscen ce increase Glousescence Polariza bility op the…
A:
Q: Steroids
A:
Q: Part 2 What is he molar mass of acetone? Pournd your ansuel 10 four significant figures. Part 3 what…
A: 2) Given: Acetone i.e. CH3COCH3.
Q: 23)
A: THE ANSWERE IS GIVEN BELOW :
Q: Write the correct Keq expressions of the reac- tions below: 1. H20 (1) A H2 (g) + O2 (g) 2. SO2 (9)…
A: As per our company guidelines we are supposed to answer only 1st three sub-parts. kindly repost…
Q: Complain it ketule's model Suffarts blowing properties of benvine's (1) The shape and brand angles…
A: Here we have to explain the shape ,bond angle and bond length of benzene by using kekule structures…
Q: CHM220 Page 3 он m. CH3 n. HC
A:
Q: MCQ 39: Chlorine reacts with cold hydroxides ions in a disproportionation reaction and results in A.…
A: Write the reaction of Cl2 with NaOH. 2 NaOH + Cl2→NaCl + NaOCl + H2O Aqueous NaCl, NaOCl solution is…
Q: 20)
A: A solution containing 0.0209 M maleic acid and 0.0297 M disodium maleate.
Q: Common aspirin is acetylsalicylic acid, which has the structure shown below and a pK, of 3.5.…
A: HC9H7O4 <——> H3O+ + C9H7O4- Using Ka = [H3O+][C9H7O4-]/[HC9H7O4] Ka is dissociation…
Q: which of the compound with strogest dipte? a) NH3 b.) H25 ng is F.) PH 3 (.) CHOCH3 Pu) CH2 CH2
A: Dipole refers to formation of two charged poles in a molecule i.e. a partial positive part and…
Q: 72) Consider The organic mole wle xy with The following date. 2.20 & Normal F-P Normal b.P 42.62…
A: Given: Csolid=1.16 J/goCCliquid=3.89 J /goCmass=3.21 gmolar mass=65.30 g/mol
Q: Question 4 Identify A andB NO2 A ČH3 A: CH3CI & Anhy.AICI3 A B: Conc.HN03 A: CH3CI & Anhy.AlCl3 B B:…
A:
Q: 3. What are AmixG and AmixS for the combination of 1.00 mol of toluene and 1.00 mol benzene at…
A: The Gibbs free energy of mixing, ΔGmix, for the combination of 1.00 mol of toluene and 1.00 mol of…
Q: Select the hydrophilic species. A. NaCl B. PO3−4 C. C18H34O2 D. vitamin D (C27H44O) E. CH4 F.…
A:
Q: How much NaAlO2 is required to produce 1.00 kg of Na3AlF6? 6HF(g) + 3NaAlO2(s) --> Na3AlF6(s)…
A: Consider the given balanced equation is as follows; Reaction: 6HF(g) + 3NaAlO2(s) →…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- Compound A, composed of C, H, and O, is heated in a1.00-L flask to 160.C until all of the A has vaporized and dis-placed the air. The flask is then cooled, and 2.48 g of A remains.When 0.500 g of A burns in O₂, 0.409 g of H₂O and 1.00 g of CO₂ are produced. Compound A is not acidic, but it can be oxi-dized to compound B, which is weakly acidic: 1.000 g of B is neutralized with 33.9 mL of 0.5 Msodium hydroxide. When B is heated to 260C, it gives off water and forms C, whose solu-tion in CDCl₃(where D is deuterium, 2H) has one peak in its1H-NMR spectrum. (a) What are the structures of A, B, and C?(b) Compound A is a controlled substance because it is metab-olized to the weakly acidic “date rape” drug GHB, C₄H₈O₃.What are the structure and name of GHB?IUPAC name of the compound in the figureCu3P wha is the name?

