Acetyisalicylic acid commonly knowWn as Aspirin is used tó treat feve inflammation, and acts as a blood thinning agent. A group of STEM student experimented on the synthesis of this compound using recrystallizatio technique. The following are the initial materials for the experiment: 5.00 g c pure salicylic acid, 7.14 g of acetic anhydride and 8 drops concentrate sulfuric acid as catalyst. After successful synthesis, the group was able to collec 5.25 grams of Aspirin. (Atomic weights: Ċ= 12 amu; H = i amu; O= 16 amu density acetic anhydride = 1.08 g/mL) Chemical reaction involved: он E-CH, с-он C-CH CH3-C-OH -O- Acetic anhydride Acetic Acid Salicylic acid (C7H&O3) Acetylsaicyic acid (C 9H3O4) (C 4H6O3) (C 2 H402) ow many molecules will be present in the Aspirin produced? O 2.18 x1022 7.44 x1022 8.13 x1022 O 9.20 x1022 None of the above
Acetyisalicylic acid commonly knowWn as Aspirin is used tó treat feve inflammation, and acts as a blood thinning agent. A group of STEM student experimented on the synthesis of this compound using recrystallizatio technique. The following are the initial materials for the experiment: 5.00 g c pure salicylic acid, 7.14 g of acetic anhydride and 8 drops concentrate sulfuric acid as catalyst. After successful synthesis, the group was able to collec 5.25 grams of Aspirin. (Atomic weights: Ċ= 12 amu; H = i amu; O= 16 amu density acetic anhydride = 1.08 g/mL) Chemical reaction involved: он E-CH, с-он C-CH CH3-C-OH -O- Acetic anhydride Acetic Acid Salicylic acid (C7H&O3) Acetylsaicyic acid (C 9H3O4) (C 4H6O3) (C 2 H402) ow many molecules will be present in the Aspirin produced? O 2.18 x1022 7.44 x1022 8.13 x1022 O 9.20 x1022 None of the above
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter5: Alkenes: Bonding, Nomenclature, And Properties
Section: Chapter Questions
Problem 5.31P
Related questions
Question
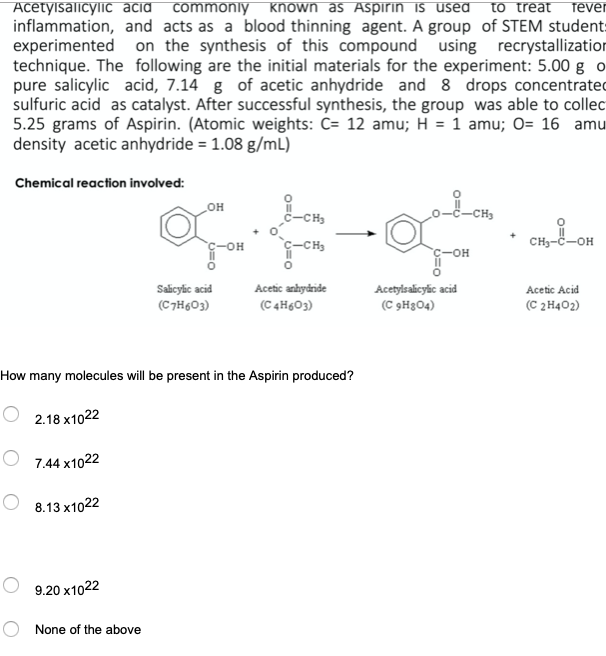
Transcribed Image Text:AcetylsalicyliC acid commonly known as Aspirin is used
inflammation, and acts as a blood thinning agent. A group of STEM student:
experimented on the synthesis of this compound using recrystallization
technique. The following are the initial materials for the experiment: 5.00 g o
pure salicylic acid, 7.14 g of acetic anhydride and 8 drops concentrated
sulfuric acid as catalyst. After successful synthesis, the group was able to collec
5.25 grams of Aspirin. (Atomic weights: C= 12 amu; H = 1 amu; O= 16 amu
density acetic anhydride = 1.08 g/mL)
to treat
fever
Chemical reaction involved:
-CH3
c-CH3
CH3-C-OH
C-OH
Salicylic acid
Acetylsalicylic acid
(C 9H3O4)
Acetic anhydride
Acetic Acid
(C7H603)
(C 4H&O3)
(C 2H402)
How many molecules will be present in the Aspirin produced?
2.18 x1022
7.44 x1022
8.13 x1022
9.20 x1022
None of the above
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning