(c) Outline how you would investigate whether BCMAP would be an effective inhibitor for the protein in competition with the molecules that the protein normally targets, and any solvent.
(c) Outline how you would investigate whether BCMAP would be an effective inhibitor for the protein in competition with the molecules that the protein normally targets, and any solvent.
Biology: The Dynamic Science (MindTap Course List)
4th Edition
ISBN:9781305389892
Author:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:Peter J. Russell, Paul E. Hertz, Beverly McMillan
Chapter4: Cells
Section: Chapter Questions
Problem 1ITD
Related questions
Question
Transcribed Image Text:Small molecules are used as inhibitors of protein action - as drugs. They most often do this
by blocking the active site within the protein. Potential drugs can be screened
computationally to determine if they are strongly bound to the protein.
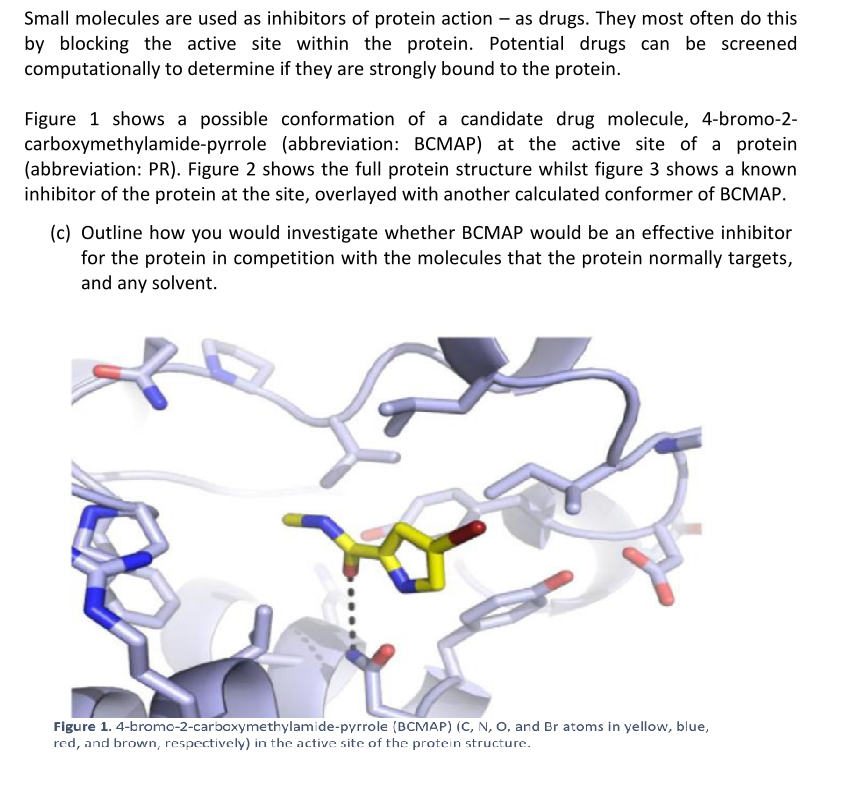
Figure 1 shows a possible conformation of a candidate drug molecule, 4-bromo-2-
carboxymethylamide-pyrrole (abbreviation: BCMAP) at the active site of a protein
(abbreviation: PR). Figure 2 shows the full protein structure whilst figure 3 shows a known
inhibitor of the protein at the site, overlayed with another calculated conformer of BCMAP.
(c) Outline how you would investigate whether BCMAP would be an effective inhibitor
for the protein in competition with the molecules that the protein normally targets,
and any solvent.
Figure 1. 4-bromo-2-carboxymethylamide-pyrrole (BCMAP) (C, N, O, and Br atoms in yellow, blue,
red, and brown, respectively) in the active site of the protein structure.
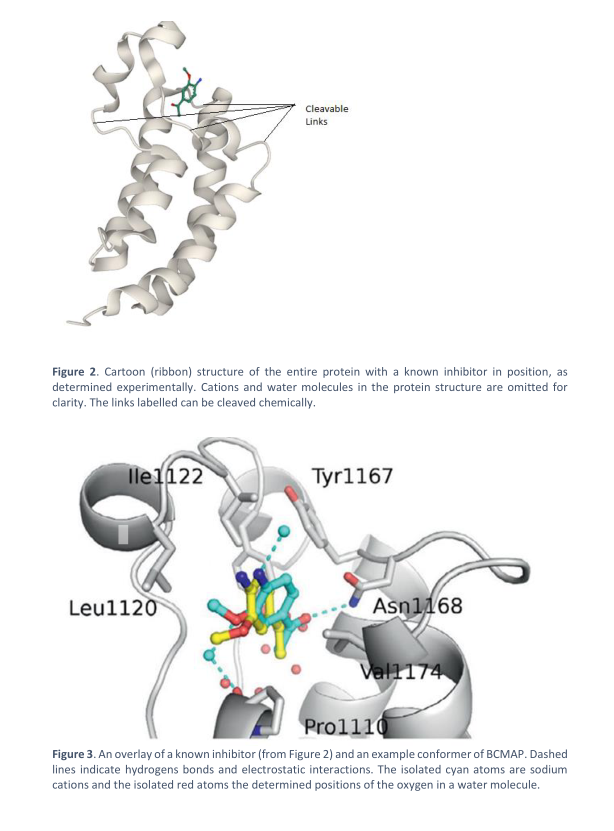
Transcribed Image Text:Figure 2. Cartoon (ribbon) structure of the entire protein with a known inhibitor in position, as
determined experimentally. Cations and water molecules in the protein structure are omitted for
clarity. The links labelled can be cleaved chemically.
lle1122
Cleavable
Links
Leul120
Tyr1167
Asn1168
Val1174
Prol110
Figure 3. An overlay of a known inhibitor (from Figure 2) and an example conformer of BCMAP. Dashed
lines indicate hydrogens bonds and electrostatic interactions. The isolated cyan atoms are sodium
cations and the isolated red atoms the determined positions of the oxygen in a water molecule.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning