
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
in text form with proper workings and explanation for each and every part and steps with concept and introduction no AI no copy paste remember answer must be in proper format with all working!!!!!!!
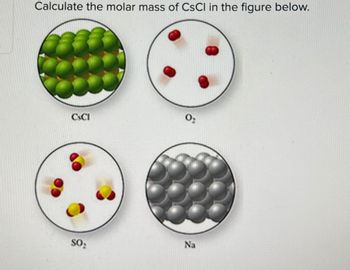
Transcribed Image Text:Calculate the molar mass of CSCI in the figure below.
CSCI
02
SO₂
Na
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by stepSolved in 2 steps

Knowledge Booster
Similar questions
- 41) please see attachedarrow_forwardW AutoSave On homewrok - Compatibility Mode - Saved - O Search (Alt+Q) raghav grover RG File Home Insert Design Layout References Mailings Review View Help O Comments A Share Draw - A A Aa v A O Find Times New Roman v 12 Normal Body Text List Paragraph No Spacing E Replace Paste В I U v ab x, x A - Dictate Editor Reuse A Select v Files Undo Clipboard s Font Paragraph Styles Editing Voice Editor Reuse Files 14. Outline a synthetic route for converting A into B. This will require several reaction steps. Show the reactant, reagents and product for each step. You do not need to show the mechanisms. OH B A Page 15 of 19 1243 words English (United States) Accessibility: Unavailable D Focus ENG 3:15 PM O Type here to search 6°F US 2021-12-16 后arrow_forwardAutoSave Off Document1 - Word P Search Savannah Hankins SH File Home Insert Design Layout References Mailings Review View Help A Share P Comments X Cut - 22.5 - A A Aav A O Find - AaBbCcDc AaBbCcDc AaBbC AaBbCcC AaB 1 Normal 1 No Spac. Heading 1 Arial B Copy Replace Paste B I U v ab x, x A- I v A - Heading 2 Dictate Editor Title S Format Painter A Select Clipboard Font Paragraph Styles Editing Voice Editor When 12.3 g of CO react with O2 according to the chemical equation: 2C0 + 02 -> 2CO2 18.0 g of CO2 are obtained. What is the percent yield of CO2 for this reaction? 10 Page 2 of 2 119 words C Focus 100% 11:26 PM O Type here to search a 99+ 20 2/3/2021 !!! lliarrow_forward
- We want to inform you that your performance as a tutor is being evaluated and will be reported to the administration. Additionally, there will be an online ranking system that not only assesses the overall performance of Bartleby as a website but also ranks individual tutors based on their question-and-answer accurate contributions. This ranking will be shared with management. Please ensure that your responses are of the highest quality, as multiple requests may be directed to the same person for assessment.arrow_forwardWe want to inform you that your performance as a tutor is being evaluated and will be reported to the administration. Additionally, there will be an online ranking system that not only assesses the overall performance of Bartleby as a website but also ranks individual tutors based on their question-and-answer accurate contributions. This ranking will be shared with management. Please ensure that your responses are of the highest quality, as multiple requests may be directed to the same person for assessment.arrow_forwardO Homework in Chem101 - due Su X My Questions | bartleby ( Periodic Table – Royal Society of how to take a screenshot on an + A learn.maricopa.edu/courses/1132256/modules/items/19018052 CHM151 17003 > Modules > Weeks 8 & 9 - Chapter 7 > Homework in Chem101 - due Sunday night CG 2020 FALL CRED Question 38 of Submit Account Home Complete the balanced neutralization equation for the reaction below: Announcements Dashboa |Modules HCIO:(aq) + NaOH(aq) – rd Concourse Syllabus Courses Grades O3+ D4+ Cisco Webex Groups 1 2 3 4 7 8 9. Tutoring/Learning Center Calendar Os Do Inbox (1) (g) (aq) History Na CI H. Help • Previous Next » Library 1:03 PM O Type here to search 10/18/2020 1L + 近arrow_forward
- ques 2 part carrow_forwardThis is NOT graded!arrow_forwardOrganic Chem 1 Week 5 Lab- Student.docx - Saved to this PC P Search shiku24 Design Layout References Mailings Review View Developer Help A A Aa v Ao E - E MaBbCcDd AaBbCc Dd AaBbCc AaBbCcD AaBi 1 Normal 1 No Spac. Heading 1 * A. P. A 三。、。 Reola Heading 2 T'tle > Select Paragraph Stylrs Editing 4. Prodiet the producti s) of the lollowing reactions, including stereochemistry when necessury und identify the mechanism of each substitution eaction (Syl vs SN2). product(s) type of reaction Br NaSMe DMSO Nal acetone HO Toxt Predicliong: On Accesiby. Irvestigate Enylish (United States) (99+ o search Pr ** FI1 FS & %23 6 7 2 3 R T. K L J F G C V Alt Alt LEarrow_forward
- View History Bookmarks Tools Window Help Firefox File Edit Module 11 HW Chapter X w NWP Assessment PlayeX Module 10 HW Chapter my.CSMD edu Module 9 HW ChapterX WNWP Assessment Play X O 8 https://education.wiley.com/was/ui/v2/assessment-player/index.html?launchld=9010073f-2141-4e90-8e6b-01c4c1518d8f#/question/5 + Module 11 HW Chapter 20 - Selected Exercises -/1 E Questio Question 6 of 8 Propose a plausible mechanism for the following transformation. Question Molecular Dra For the mechanism, draw the curved arrows as needed. Include lone pairs and charges in your answer. Do not draw out any hydrogen explicitly in your products. Do not use abbreviations such as Me or Ph Question 2 Molecular Draw -OH Question 3 Molecular Draw Pyridine Question 4 Multipart Step 1 Question 5 Multiple Choice Draw Step 1 of the mechanism. Viewing Ques Multistep HO Question 7 Multistep / Edit Drawing Question 8 Molecular Drawing MacBook Air esc ** F2 F3 F4 23 $ & * 3 8 Q W E R T Y lab S G caps lock C V hift alt…arrow_forwardpls show the process, thank you. I have tried two other options and they have not workedarrow_forward10,11arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY