E = Arial BIUA 14 %3D O DE 1E E - E E EX Format op 5 111I 11 12 ISOTOPE PRACTICE Drop and drag the electrons. 手 On the blank Bohr Models, create a model for Sodium-23 and Sodium-25. When finished, describe the difference between to the two models. (Why do they look the same? Why do they look different?) 一 ALL DONE!!! notes SLIDESMANIA.COM
E = Arial BIUA 14 %3D O DE 1E E - E E EX Format op 5 111I 11 12 ISOTOPE PRACTICE Drop and drag the electrons. 手 On the blank Bohr Models, create a model for Sodium-23 and Sodium-25. When finished, describe the difference between to the two models. (Why do they look the same? Why do they look different?) 一 ALL DONE!!! notes SLIDESMANIA.COM
Chapter7: Atomic Structure And Periodicity
Section: Chapter Questions
Problem 2ALQ: Defend and criticize Bohrs model. Why was it reasonable that such a model was proposed, and what...
Related questions
Question
Could you answer this question?
Transcribed Image Text:E = Arial
BIUA
14
%3D
O DE 1E E - E E EX Format op
5 111I
11
12
ISOTOPE PRACTICE
Drop and drag the electrons.
手
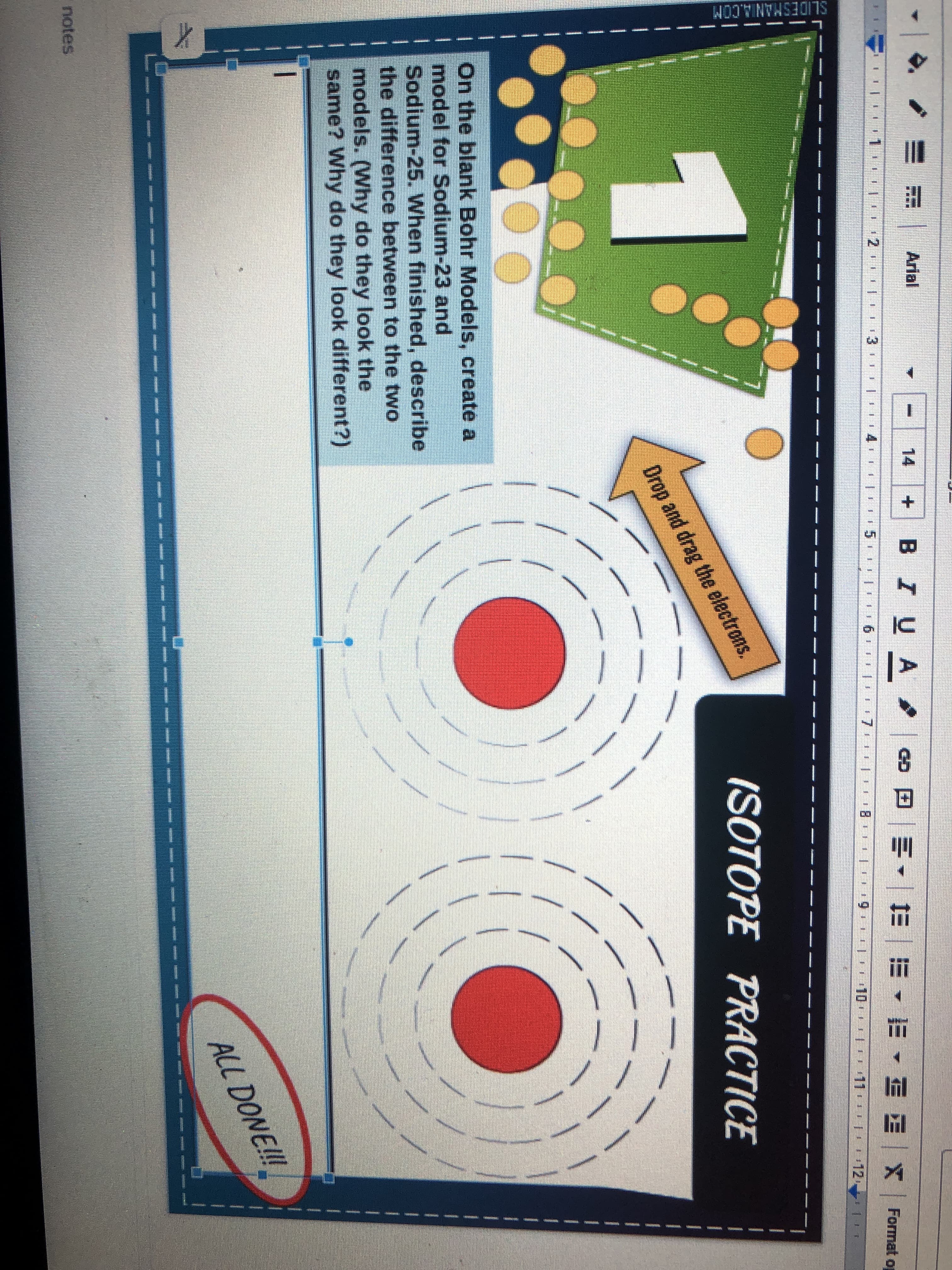
On the blank Bohr Models, create a
model for Sodium-23 and
Sodium-25. When finished, describe
the difference between to the two
models. (Why do they look the
same? Why do they look different?)
一
ALL DONE!!!
notes
SLIDESMANIA.COM
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning