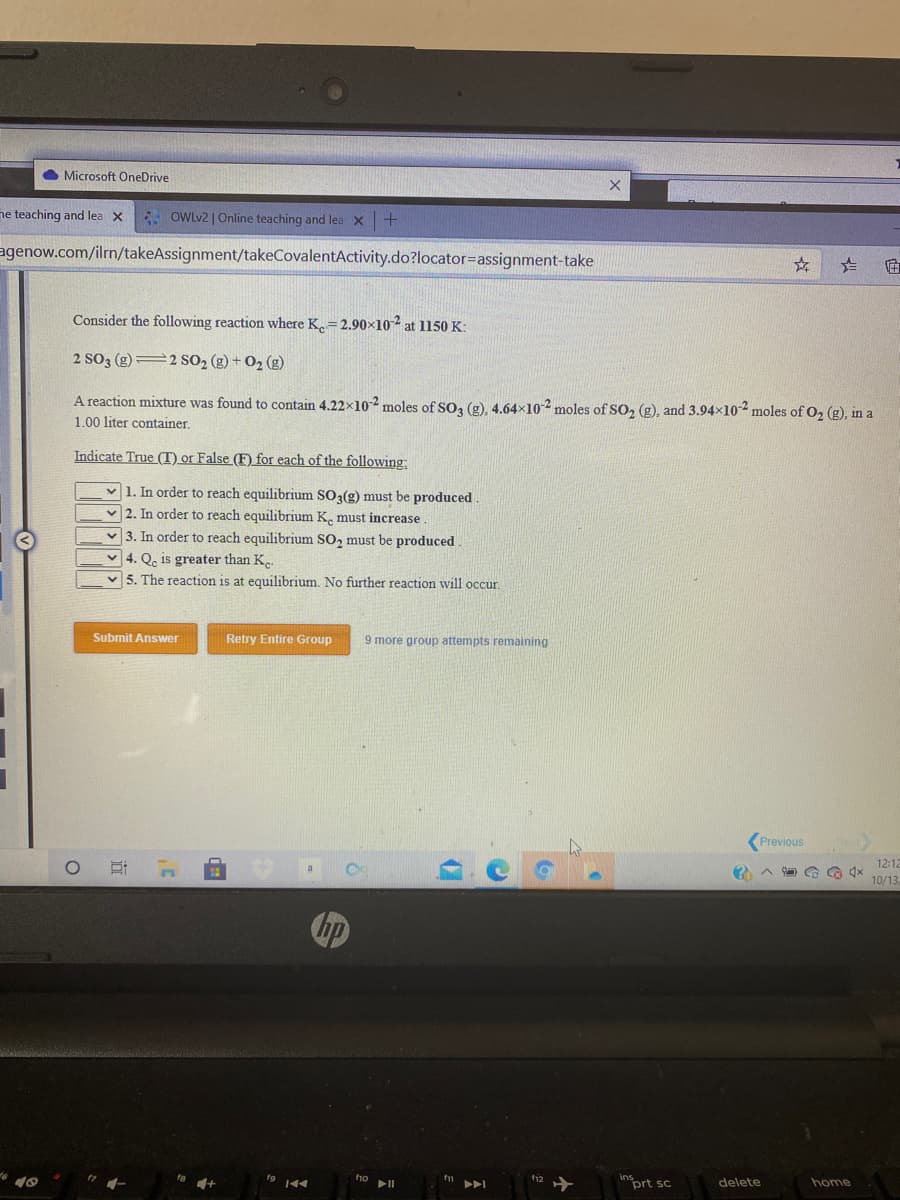
Consider the following reaction where K.=2.90×102 at 1150 K: 2 SO3 (g) 2 SO2 (g) + O2 (g) A reaction mixture was found to contain 4.22x102 moles of SO3 (g), 4.64x102 moles of SO, (g), and 3.94x10 moles of O2 (g), in a 1.00 liter container. Indicate True (T) or False (F) for each of the following: v 1. In order to reach equilibrium SO3(g) must be produced. v 2. In order to reach equilibrium K. must increase. v 3. In order to reach equilibrium SO2 must be produced. v 4. Qe is greater than K.. 5. The reaction is at equilibrium. No further reaction will occur.
Consider the following reaction where K.=2.90×102 at 1150 K: 2 SO3 (g) 2 SO2 (g) + O2 (g) A reaction mixture was found to contain 4.22x102 moles of SO3 (g), 4.64x102 moles of SO, (g), and 3.94x10 moles of O2 (g), in a 1.00 liter container. Indicate True (T) or False (F) for each of the following: v 1. In order to reach equilibrium SO3(g) must be produced. v 2. In order to reach equilibrium K. must increase. v 3. In order to reach equilibrium SO2 must be produced. v 4. Qe is greater than K.. 5. The reaction is at equilibrium. No further reaction will occur.
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 12.ACP
Related questions
Question
Transcribed Image Text:Microsoft OneDrive
ne teaching and lea x OwLv2 | Online teaching and lea X +
agenow.com/ilrn/takeAssignment/takeCovalentActivity.do?locator=assignment-take
Consider the following reaction where K.=2.90×102 at 1150 K:
2 SO3 (g) 2 SO2 (g) + O2 (g)
A reaction mixture was found to contain 4.22x102 moles of SO3 (g), 4.64×10-2 moles of SO, (g), and 3.94x102 moles of O, (g), in a
1.00 liter container.
Indicate True (T) or False (F) for each of the following:
v 1. In order to reach equilibrium SO3(g) must be produced
v 2. In order to reach equilibrium K. must increase
v 3. In order to reach equilibrium SO, must be produced
v 4. Qe is greater than K..
v 5. The reaction is at equilibrium. No further reaction wvill occur.
Submit Answer
Retry Entire Group
9 more group attempts remaining
Previous
Ne
12:12
10/13
hp
112
inort sc
delete
home
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning