Q: Advantages and disadvantages relationship of conductance and conductivity.
A: Conductance It is defined as the reciprocal of resistance of a conductor or the ability of any…
Q: Nylon is a fiber and it has some crystallinity (influences hardness). Looking at the structure for…
A: As you can see the structure of nylon 6, nylon 66 below and so from that we can determine the…
Q: write an essay explaining bonding in water cohesion.
A: Cohesion means how much molecules like to stick to itself. For water molecule there operate very…
Q: What is the best description of conductivity? Select one: O The amount of electricity present in a…
A: Conductivity of a substance is the degree to which electricity passes through substance.
Q: 2.5 Polymer/Filler Interface and Mechanical Properties Talk in general about the influence of…
A: A series of composites that may be based on the low and high density polyethylene matrices with…
Q: Theoretically, the inherent viscosity for solutions (not pure solvents) should always be a.…
A: Inherent Viscosity ηi = ln ηr/c ln ηr is zero for puresolvent and increases with increasing…
Q: Sodium phosphate (Na3PO4) is soluble in water. How would you expect its conductance to compare to…
A: The conductivity of all ions supplied by one mole of an electrolyte present in a certain amount of…
Q: describe the follwoing? are dislocations nessesary or bad for us? additionally define 3 types of…
A: It's necessary. Dislocations play an essential role in the plastic deformations of crystalline…
Q: Water samples found in the laboratory and in the environment may differ significantly in a number of…
A: Lowest Conductivity Deionized Water Fresh water Brackish water Sea water Highest Conductivity
Q: How does crystallinity affect the mechanical properties of materials.
A: Crystallinity affects the mechanical properties of materials. The thermal performances and…
Q: 5) An increase in equivalent conductance of a strong electrolyte with dilution is mainly due to : A…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Predict what group solid Gold (Au) would belong in if you tested it for conductivity in and out of…
A: When solid gold is dissolved in water, what will happen has to be predicted.
Q: Which of these graphs allows you to determine your CMC? CMC CMC In(concentration) Concentration…
A: The CMC is an important characteristics of surface tension. The surface tension changes strongly…
Q: MCQ 2: A very small amount of electrical conductivity is measured in A. silicon (Si) only B.…
A: We know that the semiconductors are used to measure the electrical conductivity and silicon and…
Q: How can solid solution occur between materials?
A: Solid solutions occur between materials as follows.
Q: Answer the following questions about KBr and BaBr2: Which has a higher molar conductivity? ISelect ]…
A: "Since there are multiple sub-parts in this question and it is not mentioned that which one has to…
Q: what will an electrical conductivity do when the size of a sample increases
A: Electrical conductivity is an intensive property and does not depend on the size of the…
Q: he packing efficiency (in
A:
Q: what is liquid junction potential ?
A: When two different concentrations of electrolyte solution come in contact forms liquid junction…
Q: Compare fracture mechanisms between ductile metals and polymers
A: Ductile fracture is the most common room-temperature mechanism of failure in metals. We will reserve…
Q: Find the molar conductivity according to these data and draw a graph.
A: The formula for calculating molar conductivity is: -----(1)
Q: Es specific conductance?
A:
Q: 3. Two metals have the same size but different type. The thermal conductivity of X = 2 times the…
A: Zeroeth law of thermodynamics says that when two substances at different temperature are brought in…
Q: Variable electrovalency is shown by which block elements?
A: The distribution of electrons in atomic or molecular orbitals of an atom or of a molecule is known…
Q: Can you draw a graph of molar conductivity using the data?
A: According to Debye-Huckel-Onsager theory the molar conductivity is related to the concentration for…
Q: Why did the compounds acted differently when conductivity was tested?
A: Conductivity is basically the ability of movement of electricity or heat throughout the chemical…
Q: Talk about the reason for surfactants to reduce the surface tension.
A: The compounds which which lowers the interfacial tension or we can say that lowers the surface…
Q: Which of the ff. angles is used for turbidity measurement of a colored solution? 180 degrees O 135…
A: Please find your solution below : 90 degrees angle is used for turbidity measurement of a coloured…
Q: d) What would happen to the Rr of the spots if you increased the polarity of the mobile phase?…
A: Chromatography is an important biophysical technique that enables the separation, identification,…
Q: Q/ Briefly explain how these factors affect wettability 1-the pore size 2- the surface roughness…
A: Wettability: it is an ability of a liquid to stay in contact with a solid surface.
Q: Identify the type of bonding that occurs in each solution and determine whether they are conductive…
A: Chemical bonds can be either ionic or covalent based on whether the electrons are completely…
Q: What are the types of orientation that shrink films go through?
A: A shrink can go through one direction (unidirectional or mono-directional) or in both directions…
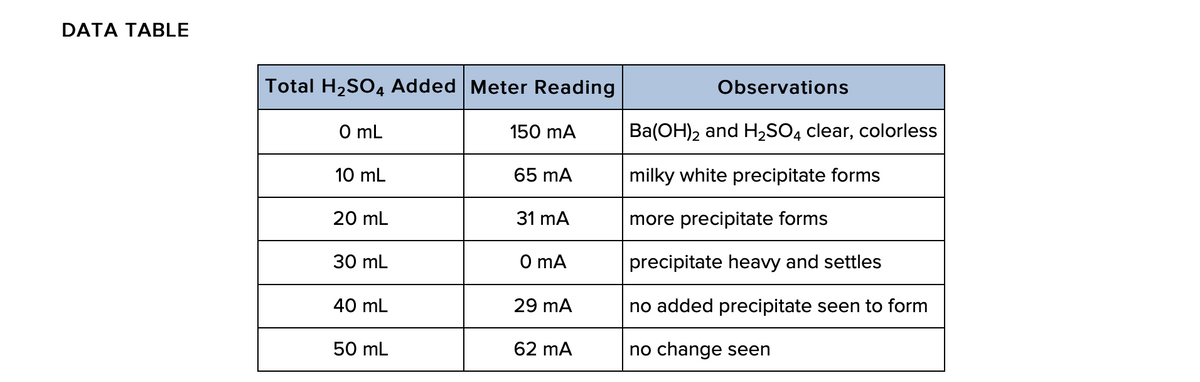
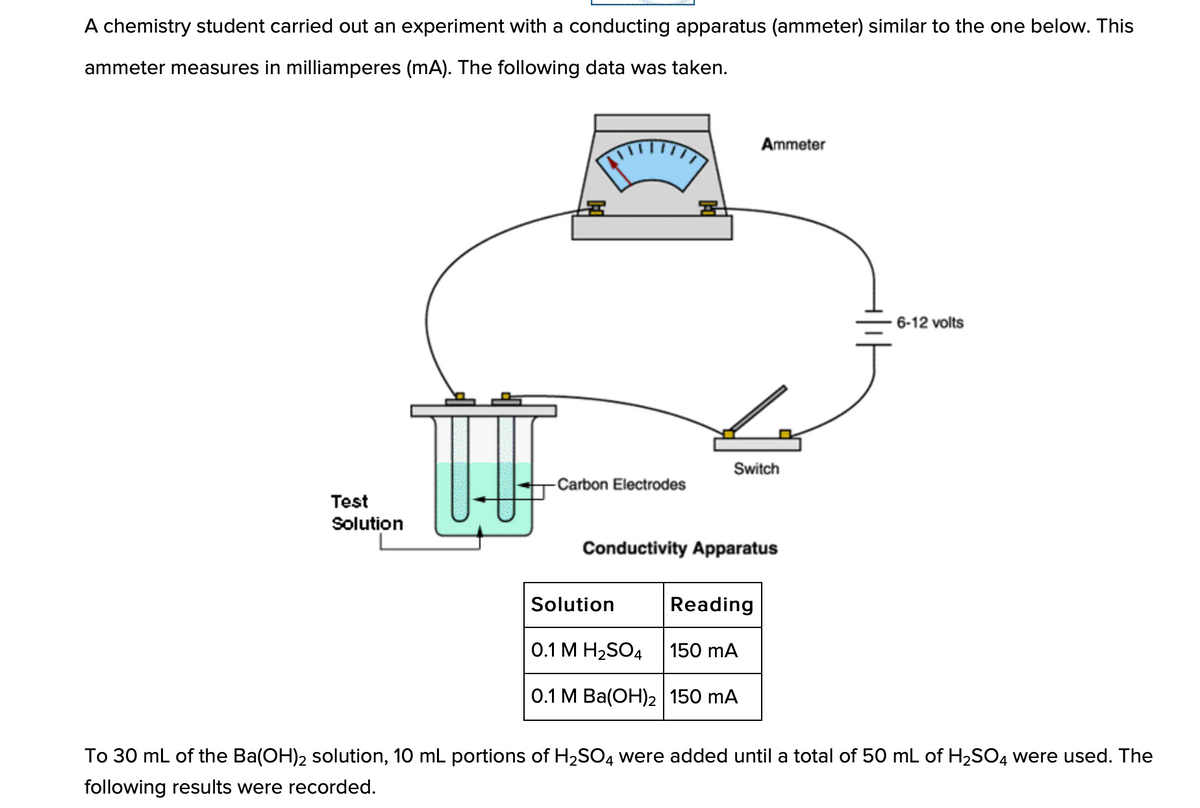
Q: When the conductivity is at a minimum, what must be true about the amount of Ba(OH)2?
A: BaOH2 dissociates as Ba2+ and 2OH- .H2SO4 dissociates as 2H+ and SO42-
Q: Alloying can harden the metals because
A: The main alloying elements which affect hardenability are carbon ,boron. Alloys contain atoms of…
Q: As G increases, the degree of mixing Increases but then decreases O Decreases O Stays the same…
A: When you add two or more gas in the container then Molecules are able to more collide to each other…
Q: ?Which statement about the term infinite dilution is correct The molar conductivity at infinite…
A: The ionic compounds are the chemical compounds that are formed of the cation and anions. When the…
Q: Which IMF is in water? 1. Hydrogen Bonding 2. London Dispersion Force 3. Dipole Force 4. Ion-Dipole…
A: As the water molecules attract each other and form bonds, water displays properties such as high…
Q: What is valance for Be
A: Electronic configuration for Be is given as ~ 1s2 2s2 Hence we can see the valence shell of Be is…
Q: In which direction along the periodic table does thermal conductivity generally increase?
A: Thermal conductivity does not show regular increase or decrease in periodic table.In every group and…
Q: What is the difference of conductance and conductivity?
A: The conductance of a component is a measurement of how easily a current can flow through the…
Q: What are the units of molar conductivity?
A: conductivity of substance is its capacity to pass electricity . Molar conductivity can be defined…
Q: Which of the following islare TRUE about molar conductivity? I. The molar conductivity provides…
A: 4th option is correct that is I, II ,III Or I.The molar conductivity…
Q: Polymorphism can exist in any crystalline material, not just molecular solids. True or false?…
A: Crystalline solids are those where a long-range order is present in the arrangement of constituent…
Q: relative viscosity
A: Ans. Option 4. Between 0 and 1. Relative viscosity can never be negative. Relative viscosity with…
Q: What are the important types of functional groups to add to a nanoparticle to actively target it?
A: Delivering nanoparticles to the targeted tissue is one of the major challenges faced in…
Q: 1. What are the principal natural processes that affect the petrophysical properties of sedimentary…
A: The main petro-physical properties of sedimentary rocks are thermal diffusivity, conductivity,…
Q: When temperature is increased by 1°c, the mobility of ions rise by. .... In conductometry solution.…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: . The molar conductivity at infinite dilution in case of NaBr, HBr and NaAc are 116.4, 415.9 and…
A: Molar conductivity: It is the parameter that is used to measure the conductivity of an electrolytic…
Does the conductivity increase or decrease?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- A sample is analyzed for chloride by the Volhard method. From the following data, calculate the percentage of chloride present:Weight of sample = 6.0000 g dissolved and diluted to 200 mLAliquot used = 25.00 mL AgNO3 added = 40.00ml of 0.1234MKSCN for back titration = 13.20ml of 0.0930Mpls complete table given the data Determination of Ksp and Molar Solubility1. Add Ca(OH)2 to 250.0 mL distilled water with stirring until equilibrium is achieved. 2. Filter the undissolved precipitate. Measure out 50.0 mL of the supernate into a 250-mL Erlenmeyer flask using a pipette.3. Add a few drops of phenolphthalein indicator and titrate with standardized HCl solution until endpoint is achieved.4. Record the volume the HCl solution used. Perform two more trials.A local company sent you their green alternative for window cleaner to be tested for percent (w/v) acetic acid content. For your experiment, you first standardized your NaOH titrant with 0.8053 g of (99.80 % purity) KHP. You used 40.60 mL of NaOH for your standardization. After that you then analyzed a 10.00 mL sample and found that you needed 43.20 mL NaOH to reach the end point. Summary of results: Standardization Sample analysis KHP Weight (g) 0.8053 g Volume of sample 50.00 mL Purity 99.80% NaOH (mL) used 33.20 mL NaOH (mL) used 40.60 mL Determine the following: Molarity of NaOH % (w/v) acetic acid
- Sources of Error Determine the relationship between the observed/apparent value (EX) VERSUS that of the true value (ET) for the quantity being sought by writing either <, >, or = on the space provided TOPIC: Measured mass of the precipitate 1. Filter paper was dried prior to filtration. EX _____ ET TOPIC: Standardization of Titrant 2. Distilled water was not equilibrated to room temperature before the preparation of NaOH titrant. EX ______ ET TOPIC: Determination of Molar Concentration of each component (Double Indicator Titration) 3. No blank correction EX ______ ETAspirin powder = 0.8110g MW of Aspirin = 180g.mol-1 Volume of 0.5N HCl consumed in back titration = 23.50mL Volume of 0.5N HCl consumed in blank titration = 44.50mL Percent purity (USP/NF) = Aspirin tablets contain NLT 90.0% and NMT 110.0% of the labeled amount of aspirin (C9H8O4) What is the calculated weight (in grams) of pure aspirin?..A 50.00 (±0.02) mL portion of an HCl solution required 29.71(±0.02) mL of 0.01963(±0.0032) M Ba(OH)2 to reach an end point with bromocresol green indicator. ? of HCL = 29.71?? ? 0.01963 ???? ??(??)2 ?? ? 2 ???? ??? ???? ??(??)2/ 50.00?? = 0.02333 ? Calculate the uncertainty of the result (absolute error).Calculate the coefficient of variation for the result.
- Knowing that Ksp of PbI2 = 10-⁷ find out if precipitate will form when mixing 100ml of 0.01 N Pb solution (NO3) 2 and 200ml of 0.01 M NaI solutionMake an schematic diagram for procedure below: B. %SO3 determination Transfer all precipitate to the filter paper avoiding any loses during the filtration procedure. One may use a rubber policeman to scrape remaining precipitate in the beaker. Wash the precipitate in the beaker 3x with hot water before transferring to the filter paper. Test the washings with 0.1 M AgNO3 Cloudiness indicates presence of chloride ions; therefore there is a need to wash more the precipitate. Carefully lift the paper out of the funnel, fold it as demonstrated by your instructor and transfer it onto the constant weighed crucible. Dry the crucible cautiously with a small flam, as instructed by your instructor. The flame should be directed at the top of the container, and the lid should be off. Avoid spattering. After drying, char the filter paper by increasing the flame temperature. The crucible should have free access to air. The lid should be kept handy to smother the filter paper in case it catches fire.…I have gotten this wrong 8 times! Please help. See screenshot attached. Thank you! A solution containing a mixture of metal cations was treated as outlined. Dilute HClHCl was added and a precipitate formed. The precipitate was filtered off. H2SH2S was bubbled through the acidic solution. No precipitate formed. The pH was raised to about 9 and H2SH2S was again bubbled through the solution. A precipitate formed and was filtered off. Finally, sodium carbonate was added to the filtered solution and no precipitate formed. What can be said about the presence of each of these groups of cations in the original solution?
- A sodium thiosulfate solution is standardized using pure copper as the primary standard. A sample of copper weighing 0.2624 g is dissolved in acid, excess KI is added, and the liberated iodine was titrated with 42.18 mL of sodium thiosulfate solution. Calculate the molarity of sodium thiosulfate solution. 2Cu2+ + 4I- → 2CuI + I2 I2 + 2S2O32- → 2I- + S4O62-15. A 300.00 mL solution of 0.00165 M A2B5 is added to a 230.00 mL solution of 0.00380 M C2D3. What is pQsp for A2D5?Pre Lab Questions: (Each answer is to be written as a complete sentence) What is the reason for washing the precipitate with water in Step 9? Define precipitate. Define filtrate. In Step 2, what is the purpose for rinsing the stirring rod? read the Procedure to answer the questions Using a balance, mass between 1.50 – 2.00 grams of sodium carbonate in a pre-massed 150mL beaker. Add 20 mL of distilled water and stir thoroughly to make sure all the crystals are dissolved. Rinse the stirring rod with a little distilled water after stirring. Using a balance, mass between 1.50 – 2.00 grams of calcium chloride dihydrate in a pre-massed 50 mL beaker. Repeat Step 2 for the solution in the 50 mL beaker. Pour the calcium chloride solution into the 150mL beaker containing the sodium carbonate solution and stir. Mass a piece of filter paper. Fold the filter paper and place it into the funnel. Wet it with a little distilled water to ensure that it is stuck to the sides of the funnel. Slowly…

