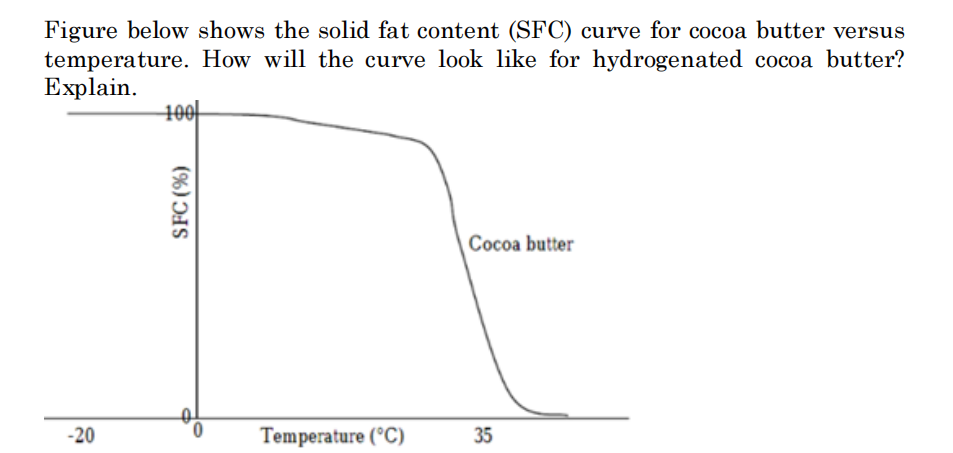
Figure below shows the solid fat content (SFC) curve for cocoa butter versus temperature. How will the curve look like for hydrogenated cocoa butter? Explain.
Q: Calculate the boiling point of a solution containing 12g glucose C6H12O6 dissolved in 200g of water…
A: Introduction Using the formula,ΔTb=Kb× molalitywhere ∆Tb is the elevation in boiling point; Kb is…
Q: Five fatty acids' classifications are shown below. Which one would you expect to have the lowest…
A: *Note: Since you have asked multiple question, we will solve the first question for you. If you want…
Q: When 50 grams of KCI is dissolved in 100 grams of water at 50 °C, the solution can be correctly…
A: Introduction: The solid substance that can be dissolved into a liquid is referred to as solute. On…
Q: Calculate the amount of rice bran (12.5% CP), corn (8.5% CP), soybean meal (49% CP) and copra meal…
A:
Q: Why is it necessary to specify the temperaturewhen making a table listing ∆G values?
A: ∆G is defined the change of Gibbs (free) energy for a system; ∆G° is the Gibbs energy change for a…
Q: Given the following data, which sample has the shortest chain fatty acids? Sample Soxhlet TBARS…
A: TBARS is defined as the content of malondialdehyde (MDA) (mg) in 1 kg of sample. TBARS analysis is…
Q: Refeeding syndrome causes dangerous fluctuations in which of the following? Question 7 options: a)…
A: Refeeding syndrome is characterized by administration of artificial feed to extremely malnourished…
Q: Solution Initial Mass (g) Final Mass (g) Change in Mass (g) % Sucrose Red 1.88 1.79 -0.09…
A: The process by which water moves from a region of high concentration to a region of low…
Q: a. Why is iodine tincture a good indicator to use for testing the presence of unsaturated fats? b.…
A: Fats are considered to be the most significant component of a balanced diet and play an important…
Q: use these in Step A belöw! Grams of carbohydrate per serving: Grams of protein per serving: Grams of…
A: Nutrients Nutrients provide energy and support growth. Calorie It is the unit of energy. It is…
Q: How much solute do you need to make 250 ml of a 3M stock of glucose?
A: The Molarity of a solution is the number of moles of a solute dissolved in a liter of solution. That…
Q: hich of the following types of oil is the best source of healthy, unsaturated fats? Group of answer…
A: ANS- Canola oil
Q: What will you observe when samples of these fats and oils are tested for unsaturation with bromine…
A: Fatty acids are building blocks of fats. Fats are one of the major biomolecules. Fatty acids can be…
Q: You have 200 mL of a 1.5 M glucose solution. How many moles of glucose are there?
A: Introduction: Glucose is a monosaccharide and a major source of energy for the living body. The…
Q: food stored for one to two hours at a temperature of 32 degrees Celsius would be safe to eat. TRUE…
A: Food storage is the most important thing as if it is not stored properly, it will lead to the risk…
Q: Name at least three test reactions for lipids. Briefly explain the the principle of each test and…
A: A group of organic compounds includes lipids that are insoluble or poorly soluble in…
Q: The average fuel value of fats is 9kcal / g . A 1 oz French fries typically contains 4.5 g of fat.…
A: All living organisms require energy to carry out cellular functions for the growth and repair of the…
Q: If the cells of your body oxidize 1 mole of glucose, by how much would the temperature of your body…
A: Specific heat capacity is defined as the amount of energy required to raise the temperature of 1 kg…
Q: Use the table below to identify the oil sample being described/ asked. Oil sample % Fatty acid by…
A: Unsaturated fatty acids are the fatty acids with one or more double bonds. Iodine is a halogen that…
Q: Which one of the following would convert a liquid fat to a solid fat:
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Refer to the table below: Oil Sample Saponification Number [A] 179 [B] 260 [C] 193 [D] 185 ✓ Which…
A: Saponification number represent the number of milligrams of KOH required to neutralize 1g of fat.…
Q: is desired to measure the acidity values of two solid fats coming to a food analysis laboratory.…
A: Since 1.2 ml KOH gets used in oil free titration it will be subtracted from volume of KOH used for…
Q: What are the hydrogenation and hydrolysis products for the triacylglycerol below? Name the starting…
A: Biomolecules are organic molecules that function inside living organisms. They are comprised of…
Q: The following has the lowest energy per gram when oxidized Carbohydrate protein ethanol O Lipids O
A: Introduction: Biomolecules are the macromolecules produced by the tissues of the organisms. They are…
Q: Protein Concentration Absorbance at 595nm (mg/mL) 0.05 0.0092 0.1 0.2 0.02 0.04 0.3 0.112 0.167 0.4…
A: There are few important points : To determine the amount of protein in an unknown sample is termed…
Q: Graph the standard curve for the starch based on this data
A: In this question, we are given concentration of starch in g/L along with the absorbance. We have to…
Q: Of the following natural fatty acids, which is predicted to have the HIGHEST melting point? Not…
A: The structure of fatty acid is correlated with a melting point. whereas unsaturated fatty acid…
Q: Using your stock solution of 2 M glucose, you need to prepare the following series of glucose…
A: Stock solution are the solutions of the concentrated solutions which are diluted in order to save…
Q: Trans-oleic acid (18:1,D9) has a melting temperature of (44.5C) and cis-oleic acid (18:1,D9) has a…
A: A cis arrangement implies that when there is a double bond, the neighboring carbon atoms are on the…
Q: Can the heat capacity of water be considered small or large? What is the biological significance of…
A: Step 1 Heat capacity (Cp) is the amount of heat required to change the heat content of one mole of…
Q: One serving of a food contains 2 g of saturated fat. What % Daily Valuefor saturated fat would…
A: In biochemistry, saturated fat is defined as the single chain of fatty acids which is also called…
Q: Fat/Oil C17H35COOH C17H33COOH C17H31COOH C17H29COOH (%) (%) (%) (%) Sample A Sample B Sample C 52 44…
A: Fats are a form of macronutrients that can be obtained from the diet. They facilitate the absorption…
Q: Water has unusually high specific heat. What does this mean?
A: The question is saying that "water has unusually high specific heat".We need to determine the…
Q: What happens when you heat ice at 0°C? (Section B C)? What is this added heat being used for?
A: You have asked multiple questions. I will answer your first question which also has two parts.…
Q: Suppose you have 150 mL of a 2.4 M glucose solution. How many moles of glucose do you have
A: Molarity is a term to define the concentration of a solution.
Q: Triacylglycerol A contains 53% oleic acid, 29% linoleic acid, 9% palmitic acid, 4% stearic acid, 1%…
A: Fats are a form of macronutrients that can be obtained from the diet. They facilitate the absorption…
Q: Carbohydrates and lipids are composed of the same chemical element, but in different proportions.…
A: Carbohydrates and lipids are the primary energy source for metabolic process in the body.
Q: Give and explain the principles of the following tests for lipids: Test for Surface Tension…
A: Asked : Principle of Test for Surface Tension Formation of Insoluble Soap
Q: Explain the principles behind the melting curve and how this helps determine G+C content. Include an…
A: Melting curve inspection is an evaluation of the dissociation features of double stranded DNA, in…
Q: The lipid shown here is produced by certain microorganisms that live in extremely warm environments,…
A: The biological membrane is composed primarily of cholesterol, phospholipids, and sphingolipids. They…
Q: how would you prepare 600 mL of a 20% strach solution beginning with a 50% stock starch solution?…
A: For calculation of stock solution, the formula used is M1V1 = M2V2 Where the M and V refer to the…
Q: the value of the ratio form the white grape juice to sugar is
A: Grape juice is made of 70 to 80 percent of water and it contains many dissolved solids (sugars,…
Q: Which of the following is true regarding glycol Choose 1 answer:
A: Ans - it requires glucose as reactant
Q: The hydrolysis of starch was stopped when the iodine test no longer gave a blue color. Does this…
A: Starch is a complex carbohydrate, which is made up of amylose and amylopectin subunits. Since…
Q: Which of the following types of fat is required to be listed on the Nutrition Facts Panel by the U.S…
A: Answer- A.Trans fatty acid
Q: Which would have more glucose, a scoop of mashed potatoes or a scoop of ice cream? Why?
A: Potatoes contain glucose in the form of starch. Ice cream contains glucose in the form of sucrose.…
Q: Why are food frozen at a temperature below 0°C?
A: Food gets spoiled with time. Various techniques are used for their preservation for a longer…
Q: Calculate the boiling points of a 8.50 m aqueous solution of fructose. Boiling point constants can…
A: Formula for boiling point: ∆Tb=Kbmwhere,∆Tb= change in boiling pointkb= boiling point constantm=…
Step by step
Solved in 2 steps with 1 images

- Directions: Indicate whether the given compound will yield positive (+) or negative (-) results. Acrolein's Test _________1. Glucose _________2. Sucrose _________3. Butter _________4. Canola Oil _________5. Albumin Huble's Test _________1. Glucose _________2. Sucrose _________3. Butter _________4. Canola Oil _________5. Albumina test tube contains both starch and the enzyme amalyase. after 30 minutes, thebtest for starch is negative and the test for simple sugar is positive, why?What are the 2 ways of production that will allow a product to be classified as an essential oil?: (Select all that apply) Choose at least one answer. a. hot pressed b. vacuum distilled c. steam distilled d. cold pressed
- Determine the correct check digit for the UPC of PH food prod: 1. 4-808887-03001- ?(purefoods corned beef) 2. 4-800088-11520-? (virginia spaghetti sauce)Explain why dichloromethane is a good solvent to remove the fat from the milk? Can you use organic solvent like ethanol to replace dichloromethane in this experiment? Experiment: Quantitative analysis of biological sample ingredients (Milk Analysis)For normal, daily use of essential oils for adults, Retha Nesbit of Plant Therapy recommends a __________ dilution. Choose one answer. a. 0.5% b. 2% c. 3% d. 5%
- Calculate the amount of phycocyanin in Sample 1 in mg where A620=0.204 and A650=0.061, taking into account the dilution factor as per question 6, and the total volume of extract as per question 4. Note your answer to 2 decimal places Sample 1 Total Extract Volume (ml) is 45 Dilution factor is 100 A280 is 1.07 A620 is 0.221 A650 is 00.97 Specific Absorbance Ratio (SAR) A620/A280 is 0.206The table shows the composition of Nutrient Agar per liter.Suppose that you need to prepare 700 mL of Nutrient Agar. How much (in g) of each component do you need? a. Beef extract b. Sodium Chloride c. Agar d. PeptoneWhich of the following oil and fat is healthier? Why?Oil/Fat Saturated (g) Monounsaturated (g) Polyunsaturated (g)Coconut oil 13 0.7 0.3Peanut oil 2.4 6.5 4.5Olive oil - 10.3 1.3Canola oil 1 8.2 4.1Safflower oil 1.3 1.7 10.4Lard 5.1 5.9 1.5Butter 9.2 4.2 0.6
- What type of error will occur if starch is added too early in the Winkler analysis? A. Positive error since the starch will turn clear after the endpoint. B. Negative error since the starch will turn clear before the endpoint. C. Indeterminate error since the starch can turn clear before or after the endpoint D. No error will occur since the starch is a stable indicatorWhich of the following is a fermentation of cheese? A. Lactic acid B. Ethanol C. Pyruvic acid D. Acetic acidWhat is it mean: tempering? Select one: a. dissolution b. pre-crystallization c. melting of sucrose d. continuous mixing What is the temperature of the candy thicken (concentration)? Select one: a. 150-155 oC b. 110-115 oC c. 140-145 oC d. 120-125 oC Which is the size limit of the wheat flour? Select one: a. less than 350 µm b. less than 250 µm c. less than 300 µm d. less than 200 µm









