Material Sample 1 50.00ml Initial volume of wator (ml) 60.00ml Final volume of water (ml.) Volume of solid (ml.) Mass, m (a) 25.95g Density, D (experimental) (giml) Density, D (acoepted) (g/ml) % Error Data Table D: Density of a Liquld Unknown1 Unknown2 10.00ml 10.00ml Volume of liquid (mL) 50.00g 50.00g Mass of empty beaker (0) 60.10g 57.75g Mass of beaker with liquid (g) Mass of liquid (g) Density, D (experimental) (giml) Material Omit Omit Density Daccepted) (giml)
Material Sample 1 50.00ml Initial volume of wator (ml) 60.00ml Final volume of water (ml.) Volume of solid (ml.) Mass, m (a) 25.95g Density, D (experimental) (giml) Density, D (acoepted) (g/ml) % Error Data Table D: Density of a Liquld Unknown1 Unknown2 10.00ml 10.00ml Volume of liquid (mL) 50.00g 50.00g Mass of empty beaker (0) 60.10g 57.75g Mass of beaker with liquid (g) Mass of liquid (g) Density, D (experimental) (giml) Material Omit Omit Density Daccepted) (giml)
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
ChapterA1: Evaluation Of Analytical Data
Section: Chapter Questions
Problem A1.22QAP
Related questions
Question
i need the answer quickly
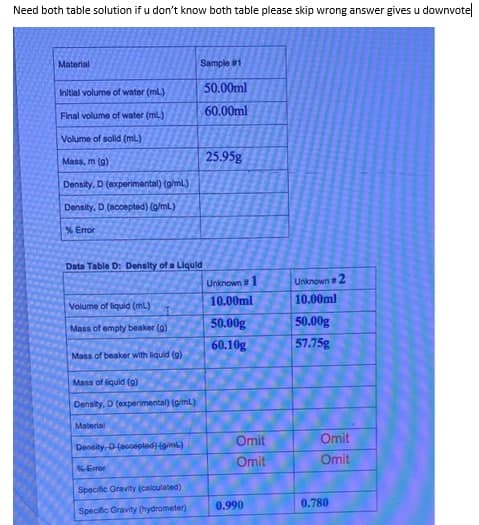
Transcribed Image Text:Need both table solution if u don't know both table please skip wrong answer gives u downvotel
Material
Sample 1
50.00ml
Initial volume of water (ml)
60.00ml
Final volume of water (ml.)
Volume of solid (ml)
25.95g
Mass, m (0)
Density, D (experimental) (g/ml)
Density, D (accepted) (g/mL)
% Error
Data Table D: Density of a Liquld
Unknown1
Unknown 2
10.00ml
10.00ml
Volume of liquid (ml)
50.00g
57.75g
50.00g
Mass of empty beaker (0)
60.10g
Mass of beaker with liquid (g)
Mass of liquid (a)
Density, D (experimental) (giml)
Material
Omit
Omit
Density D(accepted) (giml)
Omit
Omit
Error
Specific Gravity (calculated)
0.990
0.780
Specific Gravity (hydrometer)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

