o This diagram below shows the results of a simple paper chromatography experiment to identify the pen used to write a message. M comes from the ink used to write the message, and 1, 2 and 3 are from three possible pens that might have been used. Which pen might have been used to write the message? Explain why. height reached by the solvent: the "solvent front o This technique can be also used to identify particular amino acids in a mixture of amino acids. However, these are all colourless. You can dry the paper and then spray it with something like ninhydrin to make the spots visible. A mixture of amino acids (M) was tested against five known amino acids (1 to 5) and the following chromatogram was made: What amino acids were contained in the mixture M?
o This diagram below shows the results of a simple paper chromatography experiment to identify the pen used to write a message. M comes from the ink used to write the message, and 1, 2 and 3 are from three possible pens that might have been used. Which pen might have been used to write the message? Explain why. height reached by the solvent: the "solvent front o This technique can be also used to identify particular amino acids in a mixture of amino acids. However, these are all colourless. You can dry the paper and then spray it with something like ninhydrin to make the spots visible. A mixture of amino acids (M) was tested against five known amino acids (1 to 5) and the following chromatogram was made: What amino acids were contained in the mixture M?
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter26: An Introduction To Chromatographic Separations
Section: Chapter Questions
Problem 26.22QAP
Related questions
Question
Please answer both. Thank you.
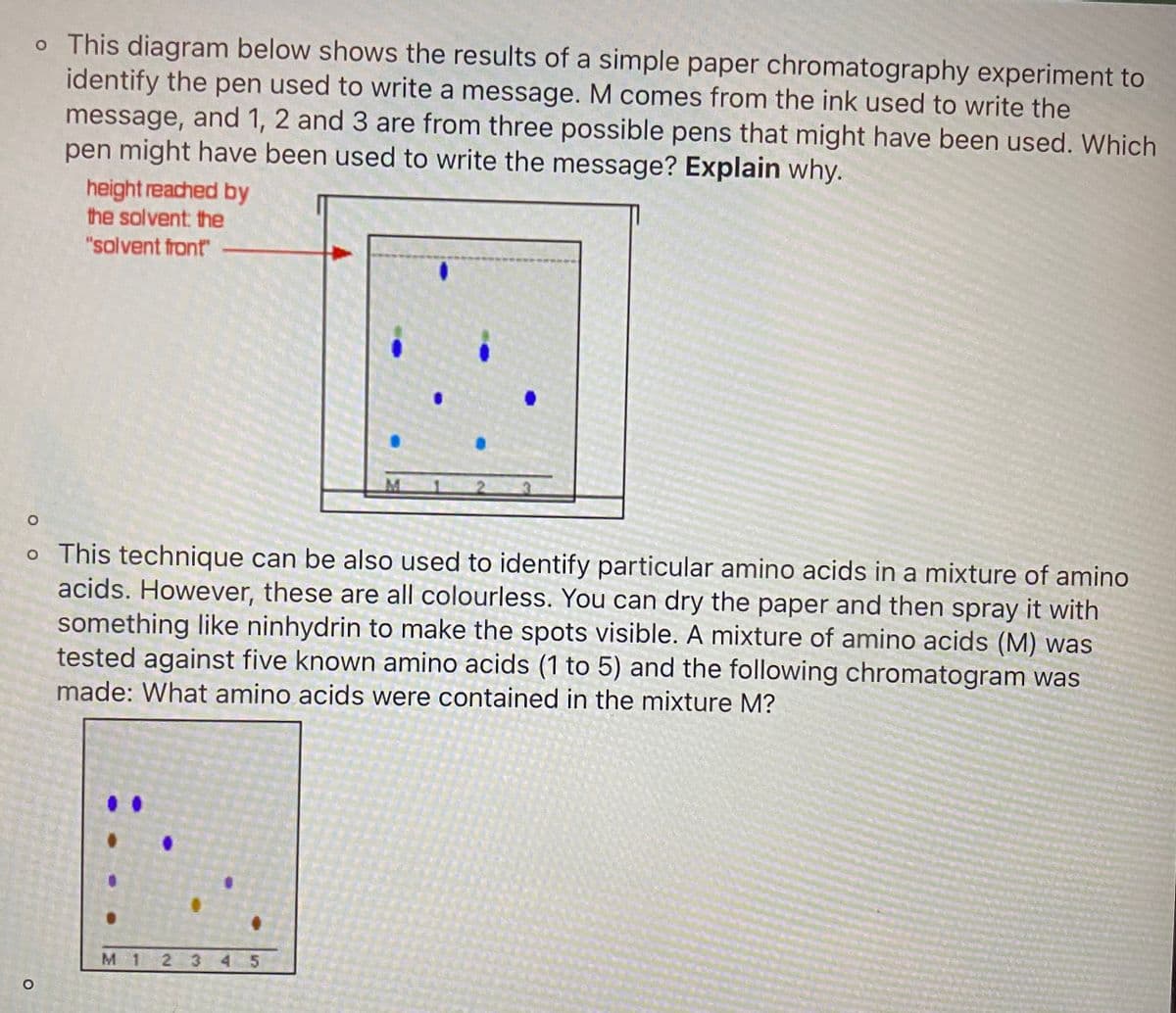
Transcribed Image Text:o This diagram below shows the results of a simple paper chromatography experiment to
identify the pen used to write a message. M comes from the ink used to write the
message, and 1, 2 and 3 are from three possible pens that might have been used. Which
pen might have been used to write the message? Explain why.
height reached by
the solvent: the
"solvent front
o This technique can be also used to identify particular amino acids in a mixture of amino
acids. However, these are all colourless. You can dry the paper and then spray it with
something like ninhydrin to make the spots visible. A mixture of amino acids (M) was
tested against five known amino acids (1 to 5) and the following chromatogram was
made: What amino acids were contained in the mixture M?
M 1 2 3 45
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT