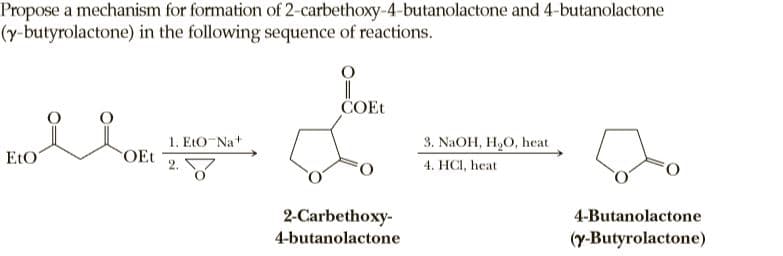
Propose a mechanism for formation of 2-carbethoxy-4-butanolactone and 4-butanolactone (y-butyrolactone) in the following sequence of reactions. COET 1. EtO-Na+ OEt 3. NaOH, H,O, heat EtO 4. HCI, heat 2-Carbethoxy- 4-Butanolactone 4-butanolactone (y-Butyrolactone)
Q: Using a Stork enamine synthesis, show how you might accomplish the following transformation: مهدديد…
A: GIVEN
Q: Treating trimethylamine with 2-chloroethyl acetate gives acetylcholine as its chloride.…
A: Trimethylamine reacts with 2-chloroethylacetate by SN2 mechanism. Thus the bond breaking and bond…
Q: Extraction (Part 2) Experiment: Separation of Neutral and basic Substances NH2 Handle aromatic…
A: The above experiment is separation of basic and neutral compounds.
Q: You lab partner mis-read the lab procedure. Instead of mixing 2-naphthol and sodium hydroxide prior…
A:
Q: Tell whether each of the following experimental errorss will raise or lower the amount of each…
A:
Q: Following are the final steps in one industrial synthesis of vitamin A acetate. H,SO, ÓH (1) (2) (3)…
A: (a)
Q: The 1H NMR spectra for two esters with molecular formula C8H8O2 are shown next. Which of the esters…
A:
Q: A synthesis of N-tosyl indole 15 starting from the pyrrole-2-carboxaldehyde 11 and acetal containing…
A: Using reaction mechanism.
Q: E. Cis-2-pentene Preamble: A student chemist in an attempt to synthesise compound B from the…
A: The given reaction from p-chlorobenzaldehyde with ammonia is a condensation reaction where water…
Q: Which reaction is NOT appropriate for the synthesis of 2-pentanone? 1. ВНЗ/ТHF 2. H2O2/NAOH А. of 1.…
A: Solved in step 2.
Q: From the lists of available reagents select the one(s) you would use to in a preparation of…
A:
Q: The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted…
A: a. Hydrochloric acid protonate the R'OH group. The reaction proceeds with the elimination of water…
Q: this drug contains one or more building blocks derived from either ethylene oxide or…
A:
Q: OH OH H2N Benzoie Acid Biphenyl Benzocaine Phenol benzocaine phenol biphenyl benzoic acid | dissolve…
A: The organic compounds present in the mixture are benzocaine, phenol, biphenyl and benzoic acid.
Q: In the Gabriel synthesis of primary amines, N-potassiophthalimide is used as a source of the…
A: This is a Gabriel phthalamide synthesis reaction. Through this reaction 1° amine is produced by the…
Q: Predict the products of the reactions of the following compounds with: (1) chromic acid or excess…
A:
Q: 9. Product of the reaction of nitrobenzene with sulfuric acid in the presence of oleum at 60 °C: a)…
A: Nitration happens when one (or more) of the hydrogen atoms on the benzene ring is replaced by a…
Q: 2. Show the products you would obtain by acid- catalyzed reaction of 3-pentanone with i. methylamine…
A: We have acid catalyzed reaction of 3-pentanone with 1. Methylamine 2.dimethylamine We have to…
Q: Chapter 20: Functional Derivatives of Carboxylic Acids 20.54 Many types of carbonyl condensation…
A:
Q: Propose an easy synthesis route to obtain 2‐nitro‐4‐pentylphenol starting from bromobenzene, without…
A: The question is based on the concept of organic reactions. We have to convert the reactant into…
Q: Following is a retrosynthetic analysis for the anthelmintic (against worms) diethylcarbamazine. N.…
A: Diethylcarbamazine is an organic compound used to treat Filariasis. It is formed y treating…
Q: ČOEt 1. EtO-Na+ OEt 3. NAOH, H,O, heat EtO 4. HCI, heat 2-Carbethoxy- 4-Butanolactone…
A:
Q: A chemist wanted to synthesize the anesthetic 2-ethoxy-2-methylpropane. He used ethoxide ion and…
A:
Q: Which of the following starting materials and reagents would be best to produce a racemic mixture of…
A: Here we are required to find the Starting material for the synthesis of 3methyl 3 hexanol
Q: 5. The acyl group is a protecting group for amines which can be deprotected by treatment with sodium…
A:
Q: 5. Write a flow chart to show how you could separate the following pairs of compounds by using…
A: Step1:By using acid base extraction, from a given mixture separation of pure compounds depends on…
Q: Spasmolytol, as its name suggests, is an antispasmodic. Given this retrosynthetic analysis, propose…
A: Spasmolytol is an antispasmodic drug that used to relieve the pain of involuntary muscle. Salicylic…
Q: Identify the reactions needed to prepare A) (1) Acetyl chloride, (2) LIAIH4, (3) Benzyl bromide B)…
A: N-benzyl-N-methylbutylamine from 1-aminobutane can be synthesized by using = B) (1)…
Q: Primary amines can also be prepared by the reaction of an alkyl halide with azide ion, followed by…
A:
Q: Sodium borohydride (NABH4) is a very selective reagent. Which functional groups can sodium…
A: Sodium borohydride is powerful reducing agent than lithium aluminium hydride.
Q: ) What is the name of the compound below? a) 3-oxopentanoic acid b) 1-acetopentanone c)…
A: Here we have to write the IUPAC name of the following componund whose structure is given.
Q: Give the shortest route to produce the desired products 1. cyclobutanoic acid to ethyl-cyclobutyl…
A: 1. Cyclobutanoic acid on treatment with SOCl2/pyridine give corresponding acid chloride which on…
Q: Propose a mechanism for the reaction of pentan-3-one with sodium hydroxide andbromine to give…
A: Mechanism for the reaction of pentan-3-one with sodium hydroxide andbromine to give…
Q: 90. What compound lucidin, rubiadin and munistin are belong? A) chrysacin derivatives B)…
A: A) Compound lucidin , rubiadin , munistin belongs to anthraquinone derivatives. B) Above structure…
Q: The benzyl ether group (OBn) is often used as a protecting group for OH groups dur- ing the…
A: The given chemical reaction is,
Q: this drug contains one or more building blocks derived from either ethylene oxide or…
A: Certirizine has three building blocks derived from ethylene oxide, shown below, and can be…
Q: Show the reagent needed to convert butanoic acid to its derivative of i. butanoyl chloride ii.…
A: In chemistry, reagent can be defined as the substance which is used to convert reactant into…
Q: Sodium borohydride (NABH4) is a very selective reagent. Which functional groups can sodium…
A: Sodium borohydride (NaBH4 ) is a week reducing agent.
Q: Show the steps necessary to convert benzene into p-chlorobenzonitrilie, using a diazonium salt at…
A: It is an example of electrophilic aromatic substitution
Q: Is fexofenadine chiral? If so, which of the possible stereoisomers are formed in this synthesis?
A: (1). The bromobenzene reacts with magnesium and then forms phenyl magnesium bromide. (2). Phenyl…
Q: propose a retrosynthetic analysis for the following molecules HO 4-chloro-6-methoxyquinazolin-7-ol…
A: The retrosynthesis of the given compounds are :
Q: What is the best synthesis route to prepare 2‐methyl‐1‐phenylpropan‐2‐ol starting from benzene,…
A: Benzene to 2‐methyl‐1‐phenylpropan‐2‐ol - Benzene treated with 1-chloro-2-methylpropane in…
Q: Din Diarin wants to methylate the diphenol shown below using methyl bromide. The reaction is a…
A: The explanation is given below-
Q: Preamble: A student chemist in an attempt to synthesise compound B from the aromatic aldehyde. Use…
A: Aldehyde and ketone reacts with ammonia gas because there is a partial +ve charge on carbon making…
Q: Show how to prepare N-ethylpropanamide from propanoic acid at or below room temperature .
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: Predict the products of the reaction with benzaldehyde with the following reagents? a. NABH4, then…
A: Since the question has four subparts so I have solved first three subparts for you. If you want to…
Q: Mechanism of the formation of the electrophilic nitrosonium ion
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: t the start of lab, Anthony adds vanillin to his Erlenmeyer flask, then adds sodium hydroxide, and…
A: According to the question we have a situation in a lab where an analyst ( Antony) transfers the…
Q: Propose an efficient synthesis for the following compound using the malonic ester synthesis: COOH…
A: The desired product can be obtained by reacting melonic ester with base followed by nucleophilic…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- Predict the products of the reaction of the below reagents with either acetophenone or phenylacetaldehyde. NaBH4, H3O+ HCN, KCN H2NNH2 CH3MgBr, then H3O+ NH2OH, catalytic acidThe 1H NMR spectra for two esters with molecular formula C8H8O2 are shown next. Which of the esters is hydrolyzed more rapidly in an aqueoussolution with a pH of 10?Show the products from the reaction of p-bromoaniline with following reagents: a)CH3Cl(excess) b) HCl c) CH3COCl d) CH3MgBr e) CH3CH2Cl, AlCl3
- The order of addition of the bases is important. What would have happened if you instead treated the initial mixture of benzoic acid, 2 - napthol and 1,4-dimethoxybenzene with a 1M NaOH aqueous solution instead of the saturated NaHCO3 solution? How would the separation of the compounds be affected? What would change?In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…
- In this study, the researcher compared S N 2 and E2reaction rates for four substrates. Three of the substrates had a second halogen on the bposition in the molecule. This work also compared the behavior of two nucleophiles:dianion I and II. You should read the abstract and look at Scheme 1 (p. 3082) and Table 4(p. 3086). Abstract: The gas-phase reactions of benzoate and phenolate containing dianions with a series of ‚-substitutedalkyl bromides (X-CH2CH2Br, X ) H, F, Cl, Br) have been studied in a quadrupole ion trap mass spectrometer.Branching ratios between SN 2 and E2 products were measured and rate constants were determined. The‚-halogens increase both the S N 2 and E2 rates, but the effect is greater for the latter process and thereforethese substituents lead to an increase in the amount of elimination. The kinetic data for the SN 2 reactions canbe analyzed via a two-parameter, linear free-energy relationship and the results indicate that field-effects (i.e.,electron-withdrawing…A chemist wanted to synthesize the anesthetic 2-ethoxy-2-methylpropane. He used ethoxide ion and 2-chloro-2-methylpropane for his synthesis and ended up with no ether. What was the product of his synthesis? What reagents should he have used?You have learned that ester may be hydrolyzed in aqueous base. Why does a workup using 50ml of 10% NaOH and 50g of ice not hydrolyze a nitro benzocaine derivative?
- Give good explanation What is the name of the product when you reduce 1-hydroxy-2-hexanone with sodium borohydride (NaBH4)?Give reagents necessary and show isolated intermediates for the synthesis below.What is the best set of reagents to achieve deoxygenation of 2-pentanone to pentane? A. NaClO2/NaH2PO4 B. LiAlH4, Et2O C. DIBAL-H, THF D. NH2NH2/t-BuOK, DMSO

