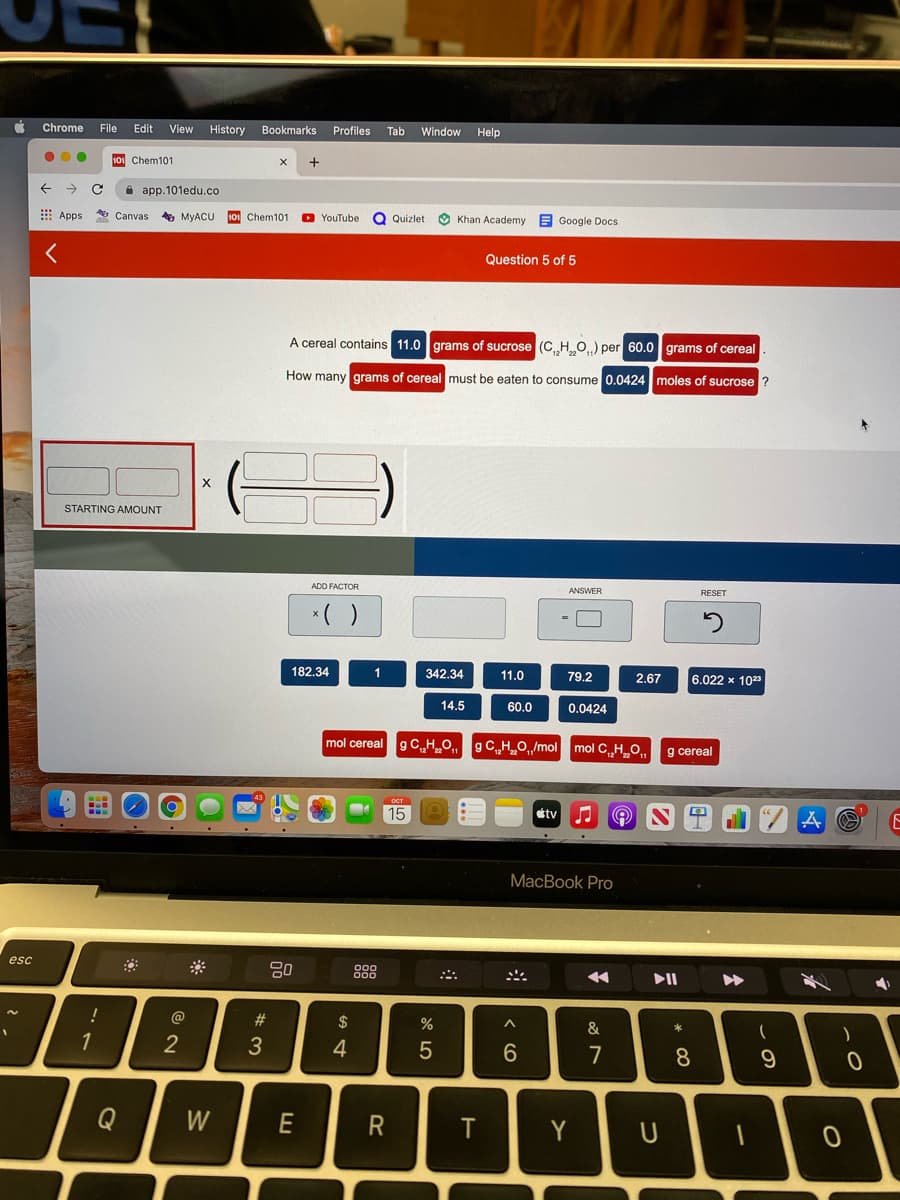
Question 5 of 5 A cereal contains 11.0 grams of sucrose (C HO) per 60.0 grams of cereal How many grams of cereal must be eaten to consume 0.0424 moles of sucrose ? STARTING AMOUNT ADD FACTOR ANSWER RESET *( ) 182.34 342.34 11.0 79.2 6.022 x 10" 2.67 14.5 60.0 0.0424 mol cereal g CH_0 gCH_O,/mol mol CH_O,, g cereal
Question 5 of 5 A cereal contains 11.0 grams of sucrose (C HO) per 60.0 grams of cereal How many grams of cereal must be eaten to consume 0.0424 moles of sucrose ? STARTING AMOUNT ADD FACTOR ANSWER RESET *( ) 182.34 342.34 11.0 79.2 6.022 x 10" 2.67 14.5 60.0 0.0424 mol cereal g CH_0 gCH_O,/mol mol CH_O,, g cereal
Chapter9: Chemical Quantities
Section: Chapter Questions
Problem 67A
Related questions
Question
Please help fill in the correct numbers and units from the below bank to solve the problem.
Transcribed Image Text:Chrome
File
Edit View History
Bookmarks
Profiles
Tab
Window
Help
Chem101
->
i app.101edu.co
E Apps
* Canvas
A MYACU
Chem101 D YouTube
Q Quizlet O Khan Academy E Google Docs
Question 5 of 5
A cereal contains 11.0 grams of sucrose (C HO.) per 60.0 grams of cereal
How many grams of cereal must be eaten to consume 0.0424 moles of sucrose ?
STARTING AMOUNT
ADD FACTOR
ANSWER
RESET
*( )
182.34
342.34
11.0
79.2
2.67
6.022 x 1023
14.5
60.0
0.0424
mol cereal gCH_O, 9CHO,/mol mol C„H O,,
g cereal
15
étv
MacBook Pro
esc
888
II
@
#
2$
%
&
*
1
2
3
4
)
6.
7
Q
W
E
R
Y
* CO
つ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div