Standard white vinegar you can buy in the grocery store is 5% concentration. That means 5% of the liquid vinegar is acetic acid and 95% of the solution is water. In a hardware store, you can buy industrial strength inegar, which is 30% concentration. This means that 30% of the vinegar is acetic acid, and the remaining 70% is water. Samuel does another experiment, this time with 5% vinegar and 30% vinegar. He sets up two science fair volcanoes (in no particular order), each with the same temperature, mass of baking soda and volume of vinegar. But one volcano uses 5% vinegar and the other volcano uses 30% vinegar. He measures the volume of gas production for the first minute of each reaction, and he records the data below. Volcano # 1 Volume of gas produced (ml.) vs. Time (s) for Volcano #1 Time Volume of gas (s) produced (ml) 60 50 25 10 20 40 38 30 46 Volume of gas produced (ml) 40 50 50 52 53 60 20 40 Time (s) 60 Volcano # 2 Volume of gas produced (ml) vs. Time (s) for Volcano 2 Volume of gas produced (mL) Time 10 10 20 30 19 30 25 26 33 39 45 Volume of gas produced m) 40 50 S 10 60 60 (u) peonpod seb jo ounon (u) peonpoad sed jo ounoA
Standard white vinegar you can buy in the grocery store is 5% concentration. That means 5% of the liquid vinegar is acetic acid and 95% of the solution is water. In a hardware store, you can buy industrial strength inegar, which is 30% concentration. This means that 30% of the vinegar is acetic acid, and the remaining 70% is water. Samuel does another experiment, this time with 5% vinegar and 30% vinegar. He sets up two science fair volcanoes (in no particular order), each with the same temperature, mass of baking soda and volume of vinegar. But one volcano uses 5% vinegar and the other volcano uses 30% vinegar. He measures the volume of gas production for the first minute of each reaction, and he records the data below. Volcano # 1 Volume of gas produced (ml.) vs. Time (s) for Volcano #1 Time Volume of gas (s) produced (ml) 60 50 25 10 20 40 38 30 46 Volume of gas produced (ml) 40 50 50 52 53 60 20 40 Time (s) 60 Volcano # 2 Volume of gas produced (ml) vs. Time (s) for Volcano 2 Volume of gas produced (mL) Time 10 10 20 30 19 30 25 26 33 39 45 Volume of gas produced m) 40 50 S 10 60 60 (u) peonpod seb jo ounon (u) peonpoad sed jo ounoA
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter10: Solutions
Section: Chapter Questions
Problem 6QAP
Related questions
Question
Transcribed Image Text:c) What data supports your answer to part (b)?
B
MacBook Air
888
esc
F2
F3
F4
F5
F6
F7
@
#
$
&
1
2
3
4
Q
W
E
R
T
Y
tab
A
S
D
F
G
caps lock
C
V
ift
control
option
command
N
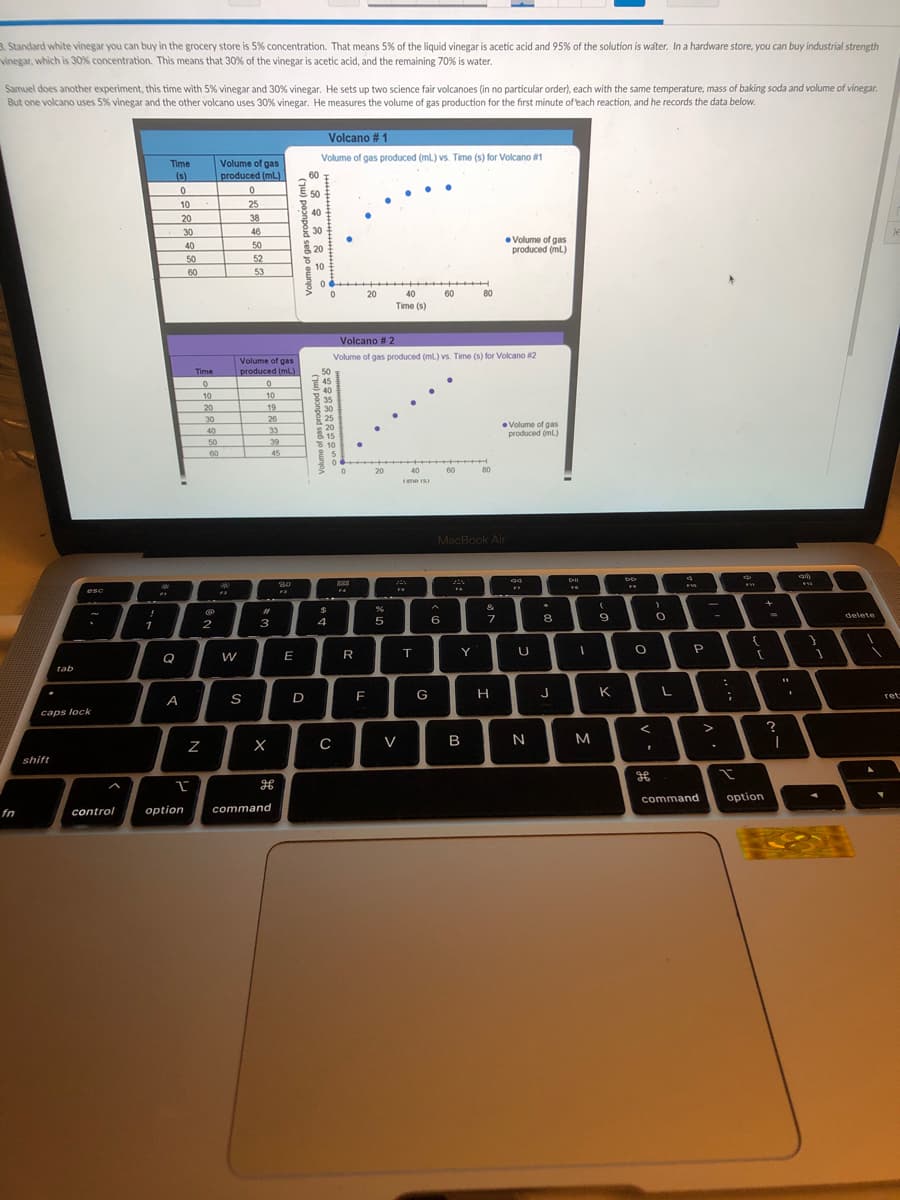
Transcribed Image Text:3. Standard white vinegar you can buy in the grocery store is 5% concentration. That means 5% of the liquid vinegar is acetic acid and 95% of the solution is water. In a hardware store, you can buy industrial strength
vinegar, which is 30% concentration. This means that 30%% of the vinegar is acetic acid, and the remaining 70% is water.
Samuel does another experiment, this time with 5% vinegar and 30% vinegar. He sets up two science fair volcanoes (in no particular order), each with the same temperature, mass of baking soda and volume of vinegar.
But one volcano uses 5% vinegar and the other volcano uses 30% vinegar. He measures the volume of gas production for the first minute of each reaction, and he records the data below.
Volcano # 1
Volume of gas produced (ml.) vs. Time (s) for Volcano #1
Volume of gas
produced (mL)
Time
(s)
60
E 50
10
25
40
20
38
30
40
46
50
30
20
• Volume of gas
produced (ml.)
50
52
53
10
60
20
40
60
80
Time (s)
Volcano # 2
Volume of gas
Volume of gas produced (ml) vs. Time (s) for Volcano #2
Time
produced (mL)
50
글 45
40
8 35
10
10
20
19
6 30
25
20
815
30
26
33
39
• Volume of gas
produced (mL)
40
50
60
45
20
40
60
80
Ime S
MacBook Air
esc
esc
&
delete
8.
1
2
3
4
E
R
Y
Q
tab
S
D
G
H
J
K
ret
A
caps lock
х
с
V
B
N
|
shift
command
option
control
option
command
fn
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning