Submit hat mass of carbon dioxide is present in 1.00 m³ of dry air at a temperature of 17 °C and a pressure of 657 torr ? Express your answer with the appropriate units.
Submit hat mass of carbon dioxide is present in 1.00 m³ of dry air at a temperature of 17 °C and a pressure of 657 torr ? Express your answer with the appropriate units.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter9: The Gaseous State
Section: Chapter Questions
Problem 37P
Related questions
Question
100%
Pls help ASAP. Pls circle the final answer and place give proper units and decimals.

Transcribed Image Text:Submit hat mass of carbon dioxide is present in 1.00 m³ of dry air at a temperature of 17 °C and a pressure of 657 torr ?
Express your answer with the appropriate units.
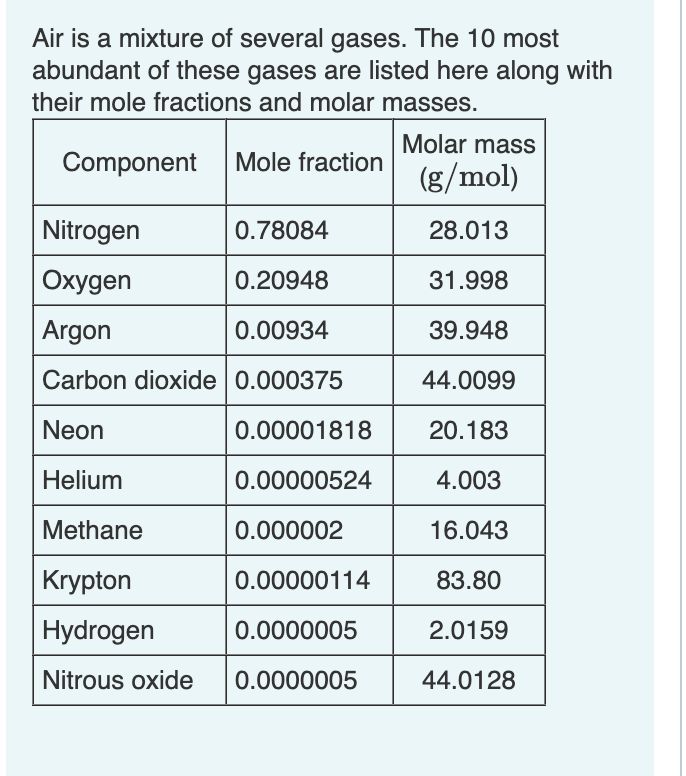
Transcribed Image Text:Air is a mixture of several gases. The 10 most
abundant of these gases are listed here along with
their mole fractions and molar masses.
Component
Mole fraction
Molar mass
(g/mol)
Nitrogen
0.78084
28.013
Oxygen
0.20948
31.998
Argon
0.00934
39.948
Carbon dioxide 0.000375
44.0099
Neon
0.00001818
20.183
Helium
0.00000524
4.003
Methane
0.000002
16.043
Krypton
0.00000114
83.80
Hydrogen
0.0000005
2.0159
Nitrous oxide
0.0000005
44.0128
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning