T UI ealil system isted Ih the first column of the table below, decide (if possible) whether the change des of the system, decrease S, or leave S unchanged. If you don't have enough information to decide, check Note for advanced students: you may assume ideal gas and ideal solution behaviour. System Change AS AS < 0 300 mL of a solution made from O AS = 0 potassium chloride (KCl) dissolved in water. 0.5 g of KCl crystallizes out of the solution, without changing the temperature. O AS > 0 not enough information AS < 0 A mixture of argon (Ar) gas and An additional 2.0 L of pure N, gas O AS = 0 nitrogen (N,) gas at 1 atm and is added to the mixture, with the O AS > 0 - 12°C. pressure kept constant at 1 atm. not enough information O AS < 0 O AS = 0 A solution made of potassium 50. mL of pure water is added to chloride (KCl) in water, at 67°C. the solution. AS > 0 not enough information
T UI ealil system isted Ih the first column of the table below, decide (if possible) whether the change des of the system, decrease S, or leave S unchanged. If you don't have enough information to decide, check Note for advanced students: you may assume ideal gas and ideal solution behaviour. System Change AS AS < 0 300 mL of a solution made from O AS = 0 potassium chloride (KCl) dissolved in water. 0.5 g of KCl crystallizes out of the solution, without changing the temperature. O AS > 0 not enough information AS < 0 A mixture of argon (Ar) gas and An additional 2.0 L of pure N, gas O AS = 0 nitrogen (N,) gas at 1 atm and is added to the mixture, with the O AS > 0 - 12°C. pressure kept constant at 1 atm. not enough information O AS < 0 O AS = 0 A solution made of potassium 50. mL of pure water is added to chloride (KCl) in water, at 67°C. the solution. AS > 0 not enough information
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter6: Equilibria In Single-component Systems
Section: Chapter Questions
Problem 6.55E
Related questions
Question
Can you tell if I am correct?
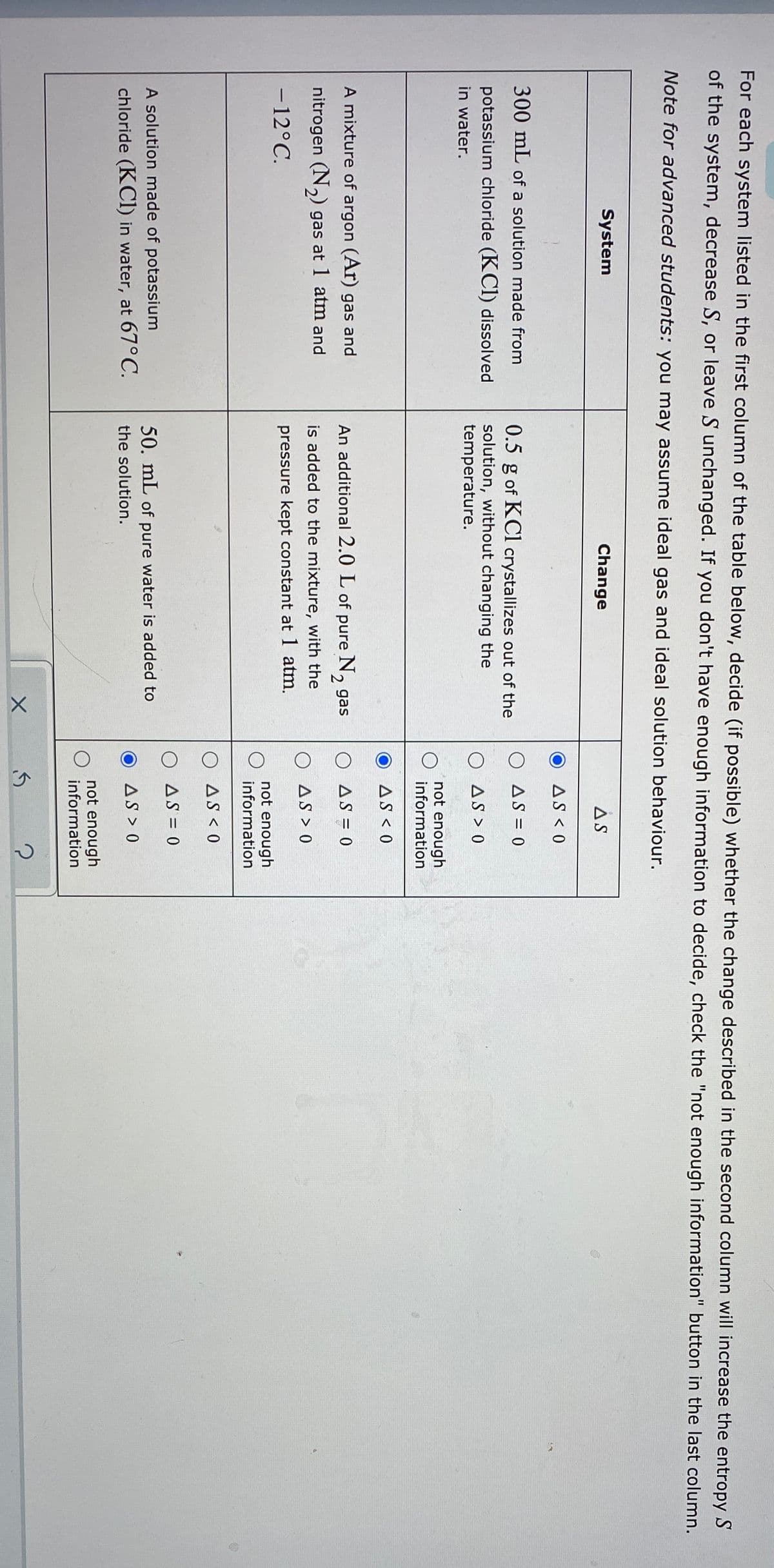
Transcribed Image Text:For each system listed in the first column of the table below, decide (if possible) whether the change described in the second column will increase the entropy S
of the system, decrease S, or leave S unchanged. If you don't have enough information to decide, check the "not enough information" button in the last column.
Note for advanced students: you may assume ideal gas and ideal solution behaviour.
System
Change
AS
AS < 0
300 mL of a solution made from
0.5 g of KCl crystallizes out of the
O AS = 0
potassium chloride (KCI) dissolved
in water.
solution, without changing the
temperature.
O AS > 0
not enough
information
AS < 0
A mixture of argon (Ar) gas and
An additional 2.0 L of pure N, gas
O AS = 0
nitrogen (N,) gas at 1 atm and
is added to the mixture, with the
O AS > 0
- 12°C.
pressure kept constant at 1 atm.
-
not enough
information
O AS < 0
O AS = 0
A solution made of potassium
50. mL of pure water is added to
chloride (KCI) in water, at 67°C.
the solution.
AS > 0
not enough
information
Expert Solution
Step 1
To check whether entropy is increasing or decreasing one just has to feel.that whether randomness is increasing or decreasing.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,
