The heat required to raise the temperature of m (kg) of a liquid from T1 to T2 at constant pressure is: -T2 Cp dT AH = m Q = In first year, you saw the equation Q = mCpAT What assumption about Cp is required to go from the first equation to the 2nd? a) b) The heat capacity (Cp) of liquid n-hexane is measured in a bomb calorimeter. A small flask (the bomb) is placed in a well-insulated vessel containing 2L of liquid n-hexane at 300K. A combustion reaction releasing 16.73KJ of heat takes place in the bomb, and the subsequent temperature rise of the system contents is measured and found to be 3.10K. In a separate experiment, it is found that 6.14 kJ of heat is required to raise the temperature of everything in the system except hexane by 3.10K. Use this data to calculate Cp (kJ/mol-K) for liquid n-hexane at 300K assuming that the condition listed in a) is valid. The density of hexane is 659 g/L.
The heat required to raise the temperature of m (kg) of a liquid from T1 to T2 at constant pressure is: -T2 Cp dT AH = m Q = In first year, you saw the equation Q = mCpAT What assumption about Cp is required to go from the first equation to the 2nd? a) b) The heat capacity (Cp) of liquid n-hexane is measured in a bomb calorimeter. A small flask (the bomb) is placed in a well-insulated vessel containing 2L of liquid n-hexane at 300K. A combustion reaction releasing 16.73KJ of heat takes place in the bomb, and the subsequent temperature rise of the system contents is measured and found to be 3.10K. In a separate experiment, it is found that 6.14 kJ of heat is required to raise the temperature of everything in the system except hexane by 3.10K. Use this data to calculate Cp (kJ/mol-K) for liquid n-hexane at 300K assuming that the condition listed in a) is valid. The density of hexane is 659 g/L.
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Chapter1: Introduction
Section: Chapter Questions
Problem 1.1P
Related questions
Question
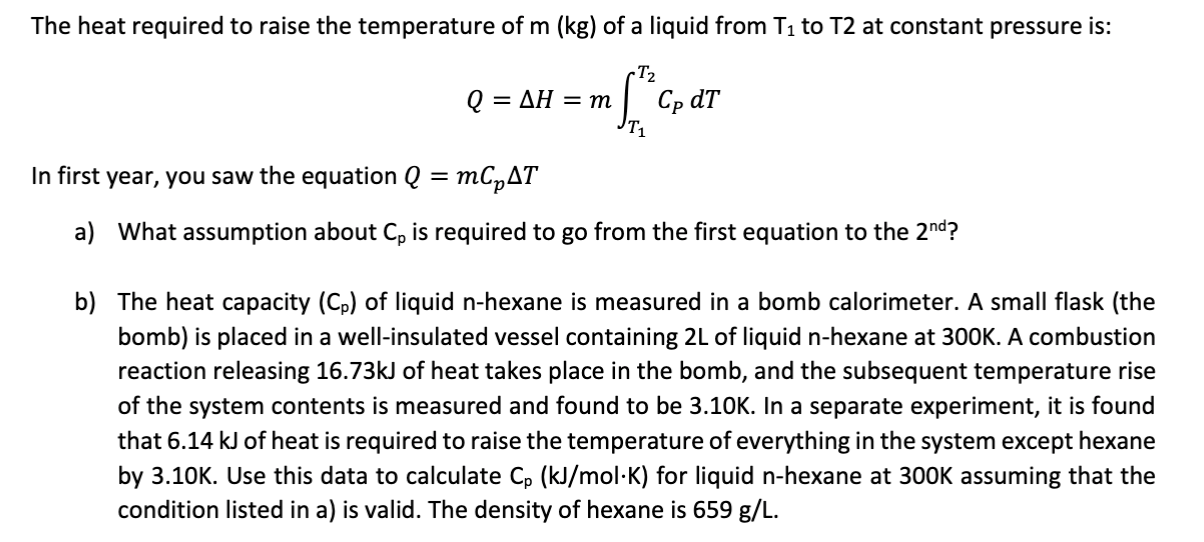
Transcribed Image Text:The heat required to raise the temperature of m (kg) of a liquid from T1 to T2 at constant pressure is:
-T2
Cp dT
AH = m
Q =
In first year, you saw the equation Q = mCpAT
What assumption about Cp is required to go from the first equation to the 2nd?
a)
b) The heat capacity (Cp) of liquid n-hexane is measured in a bomb calorimeter. A small flask (the
bomb) is placed in a well-insulated vessel containing 2L of liquid n-hexane at 300K. A combustion
reaction releasing 16.73KJ of heat takes place in the bomb, and the subsequent temperature rise
of the system contents is measured and found to be 3.10K. In a separate experiment, it is found
that 6.14 kJ of heat is required to raise the temperature of everything in the system except hexane
by 3.10K. Use this data to calculate Cp (kJ/mol-K) for liquid n-hexane at 300K assuming that the
condition listed in a) is valid. The density of hexane is 659 g/L.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Recommended textbooks for you

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:
9781285061238
Author:
Lokensgard, Erik
Publisher:
Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:
9780072848236
Author:
Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:
McGraw-Hill Companies, The