1 96% l Verizon LTE 10:23 AM Done 1. (Significant figures having a volume of 4.7 gallons exhibits a pressure of 145 psig at 70.0 °F. Using the compressibility factor chart below determine whether the contents are liquid, gas or a mixture of liquid in equilibrium with its vapor. If the contents are vapor liquid in equilibrium with its vapor, calculate the specific volume of the vapor phase in units of m'/Kg. The critical pressure of propane is 42 atm, its critical temperature is 369.9 K and its molecular weight is 44.09 g/mol important). A 20.0 lbs propane tank a or a 1.5 14 0.00 1.3 Generalized compressibility factors (z=0.27) 1.2 1.1 T. 15.00 10.00-5.00 3.00 1.0 2.00 80 09 0.9 0O 1.70 1.60 Saurated gas 150 0.8 140 1.35 0.7 1.30 1.25. 0.6 0.75 1.20 0.80 0.5 1.15 0.85 1.10 0.90 0.4 1.08 06 0.95 1.04 1.02 0.3 100 0.2 0.50. 0.1 Saturated liquid 0 0.1 1.0 3.0 4.0 5.0 10 30 0.2 0.3 04 05 2.0 20 Reduced pressure, P. X Compressibility factor, z
1 96% l Verizon LTE 10:23 AM Done 1. (Significant figures having a volume of 4.7 gallons exhibits a pressure of 145 psig at 70.0 °F. Using the compressibility factor chart below determine whether the contents are liquid, gas or a mixture of liquid in equilibrium with its vapor. If the contents are vapor liquid in equilibrium with its vapor, calculate the specific volume of the vapor phase in units of m'/Kg. The critical pressure of propane is 42 atm, its critical temperature is 369.9 K and its molecular weight is 44.09 g/mol important). A 20.0 lbs propane tank a or a 1.5 14 0.00 1.3 Generalized compressibility factors (z=0.27) 1.2 1.1 T. 15.00 10.00-5.00 3.00 1.0 2.00 80 09 0.9 0O 1.70 1.60 Saurated gas 150 0.8 140 1.35 0.7 1.30 1.25. 0.6 0.75 1.20 0.80 0.5 1.15 0.85 1.10 0.90 0.4 1.08 06 0.95 1.04 1.02 0.3 100 0.2 0.50. 0.1 Saturated liquid 0 0.1 1.0 3.0 4.0 5.0 10 30 0.2 0.3 04 05 2.0 20 Reduced pressure, P. X Compressibility factor, z
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Chapter1: Introduction
Section: Chapter Questions
Problem 1.1P
Related questions
Question
Hi this is what it can help please provide the explanation for solutions don’t worry about the exactly values please read from the graph what you can thank
Transcribed Image Text:1 96%
l Verizon LTE
10:23 AM
Done
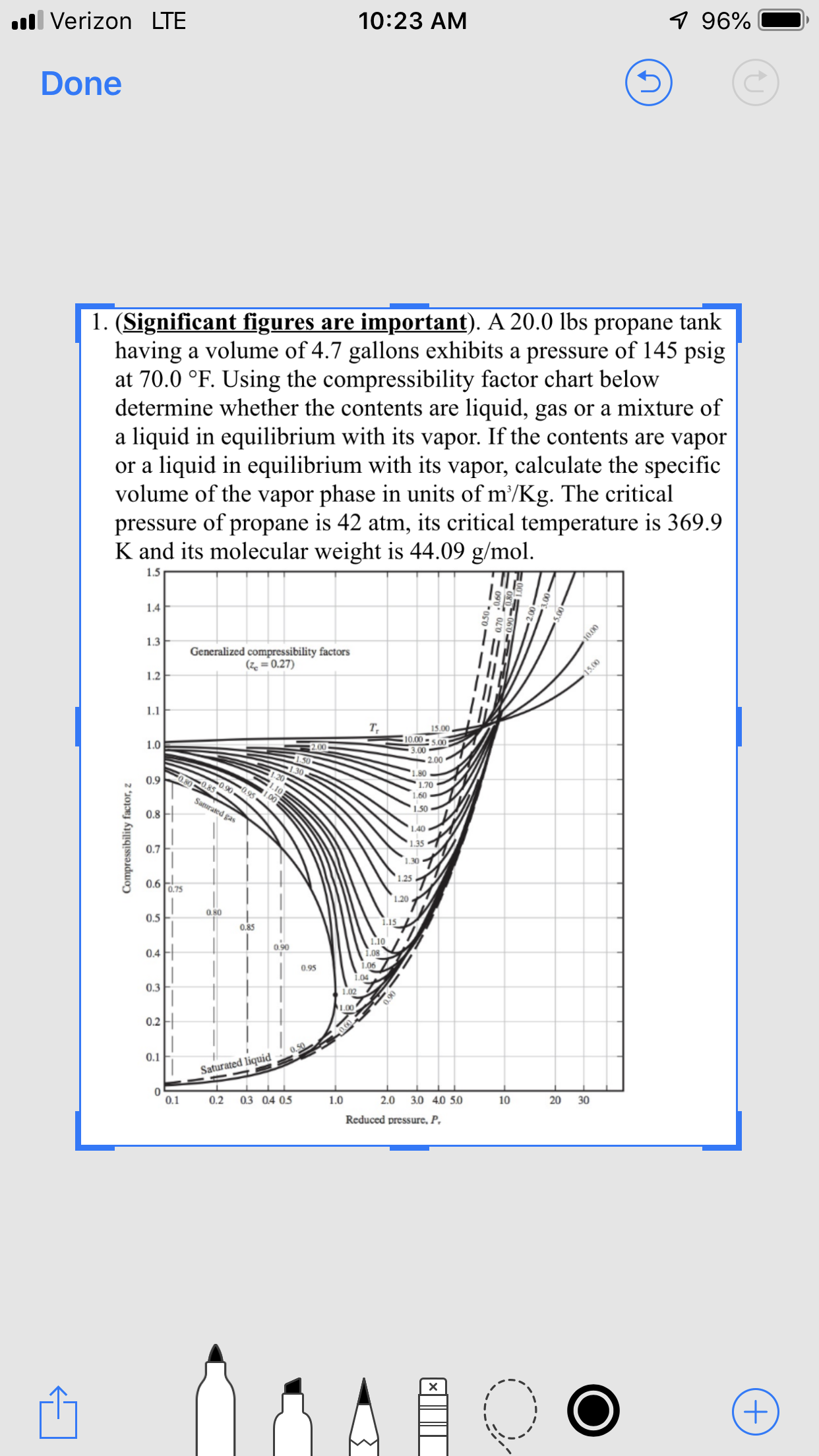
1. (Significant figures
having a volume of 4.7 gallons exhibits a pressure of 145 psig
at 70.0 °F. Using the compressibility factor chart below
determine whether the contents are liquid, gas or a mixture of
liquid in equilibrium with its vapor. If the contents are vapor
liquid in equilibrium with its vapor, calculate the specific
volume of the vapor phase in units of m'/Kg. The critical
pressure of propane is 42 atm, its critical temperature is 369.9
K and its molecular weight is 44.09 g/mol
important). A 20.0 lbs propane tank
a
or a
1.5
14
0.00
1.3
Generalized compressibility factors
(z=0.27)
1.2
1.1
T.
15.00
10.00-5.00
3.00
1.0
2.00
80
09
0.9 0O
1.70
1.60
Saurated gas
150
0.8
140
1.35
0.7
1.30
1.25.
0.6 0.75
1.20
0.80
0.5
1.15
0.85
1.10
0.90
0.4
1.08
06
0.95
1.04
1.02
0.3
100
0.2
0.50.
0.1
Saturated liquid
0
0.1
1.0
3.0 4.0 5.0
10
30
0.2
0.3 04 05
2.0
20
Reduced pressure, P.
X
Compressibility factor, z
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:
9781285061238
Author:
Lokensgard, Erik
Publisher:
Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:
9780072848236
Author:
Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:
McGraw-Hill Companies, The