Two different laboratories within a company further studied the morantel recovery percentage in shrimp when spiking with 10 ug/kg. The results from both labs are presented below. Shrimp Sample Laboratory 1 (ug/kg) Laboratory 2 (ug/kg) 108.82 113.36 2 101.59 107.47 3 102.17 109.01 a. A 4th shrimp sample was analyzed for morantel recovery percentage using laboratory 1. The result was 99.4%, can any of the data be thrown out using the Q and/or Tn test using the 95th percentile? b. Is there a statistical difference (90th percentile) between the two laboratories results in the study?
Two different laboratories within a company further studied the morantel recovery percentage in shrimp when spiking with 10 ug/kg. The results from both labs are presented below. Shrimp Sample Laboratory 1 (ug/kg) Laboratory 2 (ug/kg) 108.82 113.36 2 101.59 107.47 3 102.17 109.01 a. A 4th shrimp sample was analyzed for morantel recovery percentage using laboratory 1. The result was 99.4%, can any of the data be thrown out using the Q and/or Tn test using the 95th percentile? b. Is there a statistical difference (90th percentile) between the two laboratories results in the study?
Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
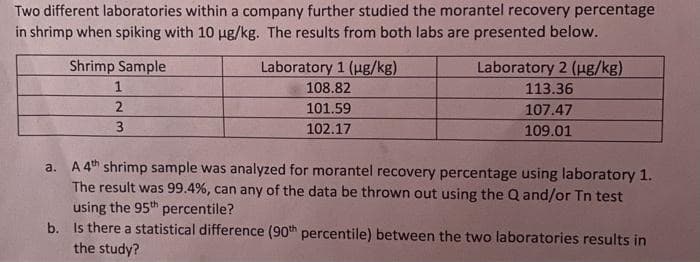
Transcribed Image Text:Two different laboratories within a company further studied the morantel recovery percentage
in shrimp when spiking with 10 ug/kg. The results from both labs are presented below.
Shrimp Sample
Laboratory 1 (ug/kg)
Laboratory 2 (ug/kg)
108.82
113.36
2
101.59
107.47
3
102.17
109.01
a. A 4th shrimp sample was analyzed for morantel recovery percentage using laboratory 1.
The result was 99.4%, can any of the data be thrown out using the Q and/or Tn test
using the 95th percentile?
b. Is there a statistical difference (90th percentile) between the two laboratories results in
the study?
Expert Solution
Step by step
Solved in 10 steps with 10 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT