V-class proton pumps run backward relative to the F-class ATP synthase. Consider the cartoon, which shows the conformations of the beta-subunits and ATPIADP + Pj of the F-class synthase. Which of the following associations between the conformation of the beta subunit and ATPIADP + P; is correct for V- Binding Change Mechanism loose binding ADP+P ATP ATP class pumps? C repeat ADP + P, ATP ATP ADP tight binding +P оpen ATP The open conformation releases ATP. Hydrolysis of ATP to ADP + Pi drives the change from tight to loose. Binding of ADP + Pj drives change from open to loose. O Hydrolysis of ATP to ADP + Pj drives the change from open to loose.
V-class proton pumps run backward relative to the F-class ATP synthase. Consider the cartoon, which shows the conformations of the beta-subunits and ATPIADP + Pj of the F-class synthase. Which of the following associations between the conformation of the beta subunit and ATPIADP + P; is correct for V- Binding Change Mechanism loose binding ADP+P ATP ATP class pumps? C repeat ADP + P, ATP ATP ADP tight binding +P оpen ATP The open conformation releases ATP. Hydrolysis of ATP to ADP + Pi drives the change from tight to loose. Binding of ADP + Pj drives change from open to loose. O Hydrolysis of ATP to ADP + Pj drives the change from open to loose.
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter20: Electron Transport And Oxidative Phosphorylation
Section: Chapter Questions
Problem 19P
Related questions
Question
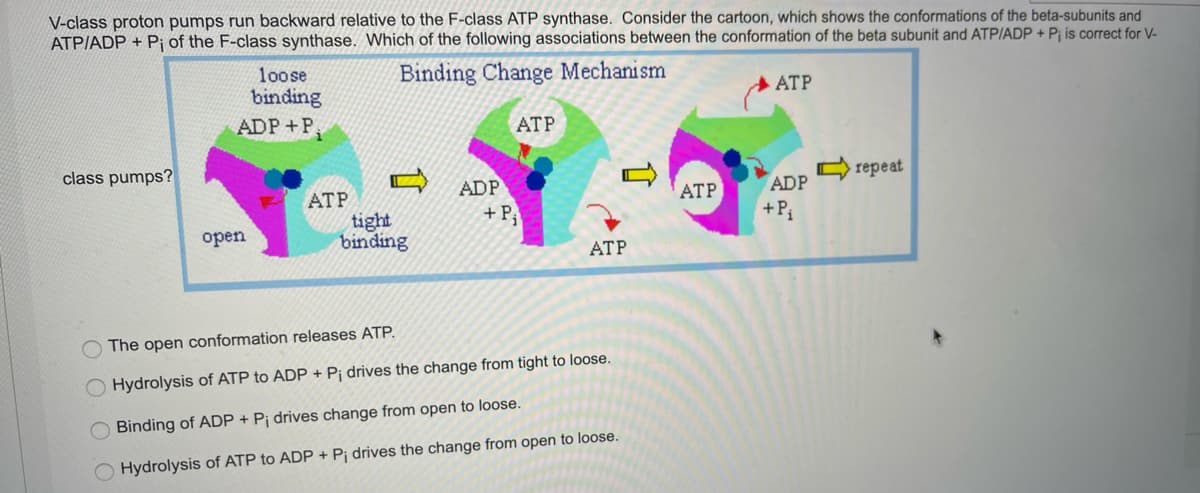
Transcribed Image Text:V-class proton pumps run backward relative to the F-class ATP synthase. Consider the cartoon, which shows the conformations of the beta-subunits and
ATPIADP + Pj of the F-class synthase. Which of the following associations between the conformation of the beta subunit and ATP/ADP + P¡ is correct for V-
Binding Change Mechanism
loose
binding
ADP+P
ATP
ATP
class pumps?
C repeat
ADP
+ P,
ADP
АТР
tight
binding
АТР
+P
оpen
АТР
O The open conformation releases ATP.
Hydrolysis of ATP to ADP + P¡ drives the change from tight to loose.
O Binding of ADP + P¡ drives change from open to loose.
Hydrolysis of ATP to ADP + Pj drives the change from open to loose.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning