Q: The number of hours per week that the television is turned on is determined for each family in a…
A: Step : 1 The required answer for the question is follow as :
Q: Solve for a, where M is molar and s is seconds. H= = (9.0 x 103 M-28-¹)(0.28 M)³ Enter the answer.…
A:
Q: What is the true volume of water delivered in A
A: To calculate the true volume of the water delivered in A = True mass of water / density of water
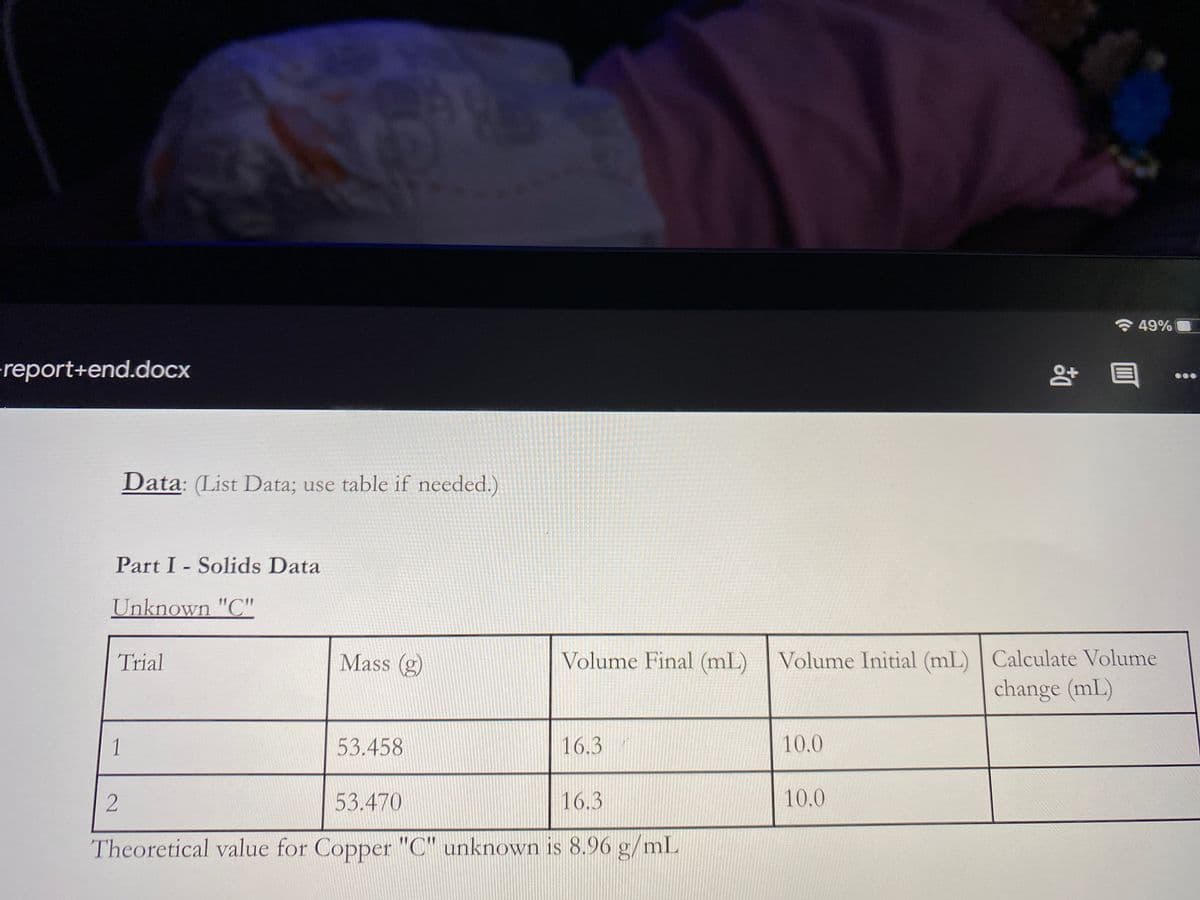
Q: Lab Data Mean Mass (g) Mean Volume (mL) Mean Density (g/mL) Slope (g/mL) 6.951 10.710 1.3 8.2 18.512…
A:
Q: Data Drawer Number Part I. Standardization of the Sodium Hydroxide Solution Mass of weighing bottle…
A: Mass of oxalic acid sample is calculated as the difference in the weight between mass of weighing…
Q: Table 1: 田 Average [NaOH) (M) Volume NaoH [NaOH) (M) Trial # KHP (g) (ml) 0.4510 22.16 0.4524 22.21…
A: The table gives mass of KHP (potassium hydrogen phthalate) and different volume of NaOH. To find:…
Q: O D. rate = k[X2] O F. rate = k[Aa[X- %3D
A: Rate law is experimental calculated expression which contains concentration of reactants raised to…
Q: Data of Candy (first trial) Mass of candy: 6.1115g Concentration of NaOH (M): 0.09639 Volume of NaOH…
A: Given: Trial 1: Volume of NaOH solution used = 17.2 mL. Trail 2: Volume of NaOH used = 17.2 mL.
Q: 1. What is the true mass of water delivered in B? A) 20.12 B) 20.13 C) 20.14 D) 20.15 E) 20.16 2.…
A: Density-ratio of mass and volume of given solution is called density. Formula is, Density is…
Q: get points. Use the References to access important values if needed for this question. 18 16 14 12…
A: The above plot is x vs y plot
Q: CHM 131 Measurements Lab ent to the coned Part 1 • In lab, the following data was recorded.…
A: Given: Measurement Measurements Estimated digit location Number of sig figures Volume of salt…
Q: Solve for x, where M is molar and s is seconds. .0 Enter the answer. Include units. Use the exponent…
A: Please find the attachment.
Q: For NH,NO3 (s) → NH,1+ (ag) + NO31- (ag Trial 1 Mass of water, g Mass of Solution, g Time, min 0.00…
A: Mass of solution = 53.84 g Mass of water = 49.34 g Mass of NH4NO3 can be calculated as follows:…
Q: B0be4141f395507aa9#10001 Academic Search I Downloads E University Librarie... UNIVERS Part A H3O*] =…
A:
Q: You are interested to know the Q10 value for mean heart rate (# of beats/10 sec) for a species of…
A:
Q: how would I start a Dixon Q test with 3 variables: Initial Volume (mL) 0.2 0.1 0.1 Final Volume…
A: Titration is the quantitative method of estimating the strength of the any component. Molarity is…
Q: 5 x 10-8 kL= ? mL be sure to show work and report correct number of sig figs and units
A: In a measurement number of digits in value are known as significant figure, all non zero values…
Q: Date Wednesday, November 20, 2019 9:59 AM 1017 100 95- 9어 85 8어 75 3277.4 7어 1448.4 65 1367.9 2936.2…
A: Some important IR peaks of 4-tert-butyl cyclohexanol are discussed below : A. Broad medium peak at…
Q: Fe(NO3)3(aq) Cu(NO3)2(aq) Submit 2 L 1 M Undo 1 L 2 M Reset Fe+ Fe3+ Fe NO, 3 L NO, NO, NO, NO,- 3…
A: The above mentioned question is explained below :
Q: b) ہنڈا F Br xxx Sug g) Br etco Fall 2021 Spring 2022 Screen Shot 2022-0...49.34 PM Organic…
A: Name the following compounds. only part f,c and g.
Q: Fri 3:26 PM istory Bookmarks Window Help A mcphs.blackboard.com anxiety statistics in high school...…
A: Given: Mass of octane= 1.10 g Temperature= 62°C Heat capacity of bomb calorimeter= 850.6 J/°C
Q: really need help calculating the molar mass using this formula setup: MM= gRT/PV = (0.016…
A: Welcome to bartleby ! We have to calculate molecular mass.
Q: Identify each transition in this flowchart as a chemical change or a physical change. 8 00 8888…
A: A question to distinguish between physical change and chemical change, which is to be accomplished.
Q: Convert into scientific notation
A: Given :- 2120000 To convert :- above value to scientific notation
Q: Run # [10;], (M) [I]. (M) [H*], (M) Product (M) Time (sec.) 1 0.005 0.050 2.00х 105 5.00 x104 9 2…
A: Since the rate law of any reaction depends only on the reactant concentrations. Hence the rate law…
Q: The following prompt will be used to answer the fırst six questions. An experiment was performed to…
A: Given: Mass of mixture is 20.100 g. Mass of initial filter paper 21.206 g. Mass of filter paper and…
Q: a- Separation is essential in chemical analysis especially when we are dealing with complex mixture…
A: We can separate the mixtures of two or more components based on different physical properties.…
Q: The following prompt will be used to answer the first six questions. An experiment was performed to…
A: Given :
Q: Table 1: Estimation of DO. burette Final SLNO Volume of water Initial sample (ml) burette Volume of…
A: Note: Since you have posted a question with multiple subparts, we will solve the first three…
Q: aye = 5.0 x 10 2 Zna+ = 1.17 x 10-5 Znya-= 3.0 x 1016
A:
Q: 21. What materials contain a precisely known concentration of a substance typically used in…
A: Reference materials are quite important for the calibration purpose in quantitative analysis. The…
Q: Time (sec) pH (RX] (H3O+] Ln[RX] 0.000 7.000 0.00002344 0.0000001 -10.6610666 15.000 6.400…
A:
Q: 3. Why is it important that all determinations be done at the same temperature?
A: 3) It is important that all determinations be done at the same temperature.
Q: Rxn: A + B -> C + D [A] M [B] M Rate M/s TRIAL 1 0.21 0.70 6.3 x 10^-3 TRIAL 2 0.63 0.65 1.89 x…
A: The rate of reaction equation for the given equation is:…
Q: Volume (Weight of sample (g) NxmEw Weight V. 100 Volume 1
A:
Q: PIH A Achieve Students: Register for X Assessment Chem 101 002 - Ac X Canvas LMS…
A:
Q: Construct a calibration curve: (a) Subtract the blank value (9.1) from all other values (this means…
A: (a) Slope = 874.05 ± 10.62 Intercept = - 29.61 ± 9.65 Calculation table vol %…
Q: Use the information below to de [Na] (M) [Onw] (M) Trial Rate (M/s) 0.10 0.10 5.0 x 10 0.20 0.10 1.0…
A:
Q: Seconds (s) Tap H2O Temp (°C) Salt Solution (+ 2.5 mL salt) Temp (°C) Salt…
A: Freezing point temperature is the temperature when liquid phase and solid phase of the substance…
Q: A typical person has an average heart rate of 69.069.0 beats/min. Calculate the given questions. How…
A:
Q: acid A student prepares a 1.2 M aqueous solution of benzoic acid (CH,CO,H). Calculate the fraction…
A: Since benzoic acid is a weak monoprotic acid. hence it will dissociate partially as C6H5CO2H…
Q: Compare the value you calculated with the actual value (-601.8 kJ/mol). Show your determination of…
A: In a reaction, for the reactant to convert to product, some amount of heat is transferred into or…
Q: Convert a speed of 799 mi/h to units of feet per minute. Also, show the unit analysis by dragging…
A: A numerical problem intended to convert mi/h into ft/min.
Q: Below is a simple set of weights obtained while immersing potato slices in various sugar solutions…
A: Weight obtained by immersing the potato slices in various sugar solution .
Q: Data Table 1. Mass of Pipetted Samples of Water (The first one is done for you. Sample Density…
A:
Q: Class A 50 mL Burette Calibration Mass of Water Delivered, g Actual Volume Delivered, Burette…
A: For the actual volume delivered we will use the concept of density of water. We know that; Density…
Q: Part 1: AHdissolution Trial 1 Trial 2 1201659 130.29 3.759 133.959 (calculated) 0.09399 (calculated)…
A:
Q: NO2 + 03 → NO3 + 02 [03], M Rate, M/s 6.30 x 10-3 1.89 x 10-2 3.15 x 10-3 Experiment [NO,],M 1 0.21…
A: From the unit of the rate constant it is obvious that its a second order reaction. So the Rate…
Q: Estimated volume of crucible 25.0 mL measured volume of crucible 23.0 mL percent error of…
A: % error=accepted value-experimental valueaccepted valuex100 %
Q: Temp °C Temp Kelvin Ka In(Ka) AG'=-RTIN(Ka) 22.1 295.25 0 o044521 -1.12 18950.34962 35 308 .1Sk -901…
A: The data given is,
Do calculations show work
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Please answer fast it’s very important and urgent I say very urgent so please answer super super fast please For the image attached For 1. a Mass of metal: Trial 1 is 35.0228 g Trial 2 is 35.0915 g Trial 3 is 34.0821 g Mass of water: Trial 1 is 20.0177 g Trial 2 is 20.0250 g Trial 3 is 20.0168 g For delta t of water: Trial 1 is 15.5 C Trial 2 is 15.7 C Trial 3 is 15.1 C For delta t of metal Trial 1 is 80.1 C Trial 2 is 80.2 C Trial 3 is 79.5 C For B my calculated Specific heat is: Trial 1 is 0.462 Trial 2 is 0.467 Trial 3 is 0.466Downvoted for wrong solution. A river is carrying water containing 2000 mg/l Magnesium Chloride into a small lake. The lake has a naturally occurring Magnesium Chloride of 50 mg/l. If the river flow is 2500 Lmin and the lake flow rate is 1.5 m³.sec¹, what is the concentration of MgCl2 in the lake after the discharge point? Assume that the flows in the river and lake are completely mixed, that the salt is a conservative substance, and the system is at steady state.Please answer super fast and answer all questions and show calculations For the image attached For 1. a Mass of metal: Trial 1 is 35.0228 g Trial 2 is 35.0915 g Trial 3 is 34.0821 g Mass of water: Trial 1 is 20.0177 g Trial 2 is 20.0250 g Trial 3 is 20.0168 g For delta t of water: Trial 1 is 15.5 C Trial 2 is 15.7 C Trial 3 is 15.1 C For delta t of metal Trial 1 is 80.1 C Trial 2 is 80.2 C Trial 3 is 79.5 C For B my calculated Specific heat is: Trial 1 is 0.462 Trial 2 is 0.467 Trial 3 is 0.466
- If 35,000 kg of whole milk containing 4% fat is to be separated in a 6-hour period into skim milk with 0.45% fat and cream with 45% fat, what are the mass flow rates of the two output streams from a continuous centrifuge which accomplishes this separation? (Ans; Cream=464.8335kg/h, Skim milk= 5368.4998kg/h)1. Estimate the density of a 25-API gravity dead oil at 100 F.calculate the flow rate in ml/hr . order 40meq/l at 10meq/hr in 1000ml in d5w supply 20meq/10ml
- can you answer, please? the experimental data are Reference: https://www.youtube.com/watch?v=OOXRkycKEOc&feature=youtu.be and https://www.youtube.com/watch?v=3wLJLm0QLpg&feature=youtu.beExpress 20,000ppb as a percentage strength A. 2% B. 0.02% C. 0.00002% D. 0.002%Chemistry The levels of an organic pollutant (P) in the groundwater at the perimeter of a plant were a cause for concern. A 10 mL sample of the water was taken and the pollutant was extracted with 95% efficiency using 25 mL of diethyl ether. GC was used to analyse the concentration of P in diethyl ether. A calibration curve was plotted for a series of standards of P which yielded the following results: Peak Area Toluene Conc. (µg/ml) 12,000 2.6 23,700 5.0 35,500 7.7 46,800 9.9 31,250 Sample Determine the concentration of P in ppb in the initial groundwater sample.
- 4. Ksp=4.1x10-36 for Pb3(AsO4)2Calculate Eofor Pb3(AsO4)2(s) + 6e- 3Pb(s) + 2AsO43-Pb2+ +2e- Pb(s) Eo= -0.126 VCALCULATE Vmax and KM values of the reaction as shown below ON LB PLOT Please don't provide handwriting solutionThere's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…



