Q: 2. What are the Electron pair arrangements for each of the molecules in #1. SiF₁ SF6 ICI,.
A: Given molecules are : a. SiF4 b. SF6 c. ICl3 What are the electron pair arrangement of each of…
Q: a laccd sign in - Sear X tab caps lock shift 1 A chemist determines by measurements that 0.080 moles…
A: Given, The moles of Iodine solid precipitates in the chemical reaction = 0.080 mole. The mass of…
Q: using any necessary reactants and reagents, propose reasonable syntheses of the following compounds…
A: The final product is formed by various steps as follow:
Q: Write the mechanism for the following aquation reaction:
A:
Q: What is the lithium carbonate concentration when 225 grams of Li₂CO3 is dissolved in enough…
A: Given mass of lithium carbonate (Li2CO3) = 225 g Molar mass of Li2CO3 = 73.891 g/mol Number of…
Q: Of the two compounds shown below, one is slightly soluble in water and the other is insoluble in…
A: Polar species likes to dissolve in polar solvent. In other words like dissolves like. Non polar…
Q: Thiophene (refer to structure below) is an aromatic compound based on Huckel's rule of aromaticity.…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Check the box under each structure in the table that is an enantiomer of the molecule shown below.…
A:
Q: A saline solution is administered intravenously to patients who cannot take fluid orally and are in…
A:
Q: .300.0 mL of 0.00325 mol/L barium chloride is added to an equal volume of 0.004 00 mol/L sodium…
A:
Q: Balanced Chemical Equation Reaction Type: At completion of reactions: Grams of nitrogen Grams of…
A: All your calculations so far are correct. N2(g) is the limiting reactant.
Q: Predict the expected major product of the reaction below? + enantiomer 1 OOOO || O IV OV HCI له…
A:
Q: Identify the substances that are oxidized and reduced, and indicate which is the oxidizing agent and…
A:
Q: Which of the following images shows a distant object moving towards Earth? The continuous spectra…
A: To answer: Which of the following images shows a distant object moving towards Earth?
Q: Roles of chemistry in agriculture, medicine, food, industry, home and water.
A: Role of chemistry in agriculture Agriculture deals with the generation of organic goods using both…
Q: ОН ОН
A: Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more prior…
Q: Write two chemical reactions that the copper chloride hydrate (Cux Cly-ztl₂0) is undergoing in this…
A: It is a hygroscopic compound. The presence of water of crystallization can be removed by heating the…
Q: A. Similar O similar overall size of amino acid O ability to hydrogen bond O same group of periodic…
A: Sulphur and selenium have similar physicochemical properties as both are members of the chalcogen…
Q: Show the calculation of the mole fraction and partial pressure of each gas in a mixture which…
A:
Q: 1. If 8.5 g of pure ammonium phosphate, (NH₂),PO,(s), is dissolved in distilled water to make 400 mL…
A: Dear student , since you have posted multiple questions we are allowed to solve only first question…
Q: Sigma bonding: A o bond arises from the straight-on overlap of two atomic orbitals. The electron…
A: Carbon 2 belongs to the carboxylic functional group which is attached to the 3 atoms and it also…
Q: How many stereoisomers does the structure below has? CH3 H- -C-F H- HC OH H- 8 16 32 -C-Br 4 -C- -CI…
A: The first question asks about the number of stereoisomers. For this we will check the number of…
Q: Potential Energy 7.01 ENDOTHERMIC AND EXOTHERMIC 7.02 ENTHALPY Reactants A Reaction Progress-…
A:
Q: What is the peroxide effect? What does it mean when there is a reversal of orientation? Detailed…
A: Markovnikovs's rule: Markovnikov' rule states that when an asymmetrical reagent is added to an…
Q: The Ksp of beryllium hydroxide, Be(OH)2, is 6.92 x 10-22. Calculate the solubility of this compound…
A:
Q: Rank the following diatomic species of oxygen in order of bond length and bond strength. A. 0₂ B.O₂+…
A: Bond strength depends upon bond order. The higher the Bond order, the higher will be bond strength.…
Q: Reactant 1 Structure Choices: SAT Structure A WAT MAT OH f Br concentrated H₂SO4 RAT Structure B OH…
A:
Q: At 25 °C, how many dissociated H+ ions are there in 389 mL of an aqueous solution whose pH is 11.71?…
A:
Q: Enter the net ionic equation for this reaction. Express your answer as a net lonic equation.…
A: Net ionic equation is an equation that represents only the molecules or ions that are actively…
Q: What are the structures of the products below? Type the letter labels of your answers. Type NONE if…
A:
Q: (a-b) (c-d) (e) 1. Br₂, H₂O mCPBA excess HCI 1. KOH 2. KCN, H,O 1. CH₂CH₂MgBr 2. Dilute H*, H₂O
A: Note - The given question is a multipart question, hence I solved first three questions according to…
Q: Select the nonpolar molecules with the lowest boiling point? A. CF4 B. CO2 C. C6F6 D.…
A: Boiling point is temperature at which vapour pressure of liquid become equal to atmospheric…
Q: Titration is a common laboratory method of quantitative chemical analysis. During the titration of…
A: Titration is volumetric method used to determine the concentration of an unknown solution by using…
Q: Consider the reactions and the pool of choices below. Type the letter labels on the blanks provided…
A:
Q: What are the coefficients of NH3 and H2O? ___ Al + ___ NO3- + ___ OH- + ___ H2O→ ___ Al(OH)4- + ___…
A:
Q: At 25 ∘C , the equilibrium partial pressures for the reaction 3A(g)+4B(g)↽−−⇀2C(g)+2D(g) were…
A:
Q: An exothermic reaction is always spontaneous. Product formation is always favored in a reaction with…
A: ΔG=ΔH-TΔS If the change in free energy of a reaction is positive , then reaction will be…
Q: In which of the following equations is the outcome wrong? O a. O b. Oc CI O d. Br. N₂+CI- NH₂ Me 1)…
A: We have to tell among the given reactions which one is wrong.
Q: 4. Indicate the organic products for the intramolecular imine formation reaction (a) and enamine…
A:
Q: Describe equilibrium constants with words and equations. For the reaction A + 2B --> 2C, the…
A:
Q: What would be the boiling point of a 9.80 m solution of NaBr an ionic compound that produces two…
A: when a solute is added to a pure solvent the boiling point of sulvent increases. This phenomenon is…
Q: [b] T(°C) Log (K) The following equilibrium data have been determined for the reaction: Persamaan…
A:
Q: ma-Q 1) NaH 2) H₂O -XX OH (c) (d) H₂N NO₂ HS. H NO₂ hint: consider that NO₂ can stabilize an…
A: This reaction follows Cyclization Reaction. In the first step Strong Base NaH abstracts proton. Then…
Q: Practice problems: Propose reasonable mechanisms for the following reactions. Use arrows correctly…
A: Here basically ring formation occurred by attacking the nucleophile on carbonyl.
Q: Enter an equation for the precipitation reaction that occurs (if any) when solutions of sodium…
A:
Q: CI™ Express your answer in the order of orbital filling as a string without blank space between…
A:
Q: A buffer solution contains 0.459 M CH3NH3Cl and 0.368 M CH3NH2 (methylamine). Determine the pH…
A:
Q: The change in entropy, Δ?∘rxn, is related to the the change in the number of moles of gas molecules,…
A: Change in number of moles in a reaction is calculated by subtracting the moles of gaseous reactants…
Q: Is the following compound aromatic, antiaromatic, nonaromatic, or is it not possible to tell?
A:
Q: Enter the complete ionic equation to show the reaction of aqueous lead(II) nitrate with aqueous…
A: Given that, there is a reaction between aqueous lead(ll) nitrate (Pb(NO3)2 with aqueous potassium…
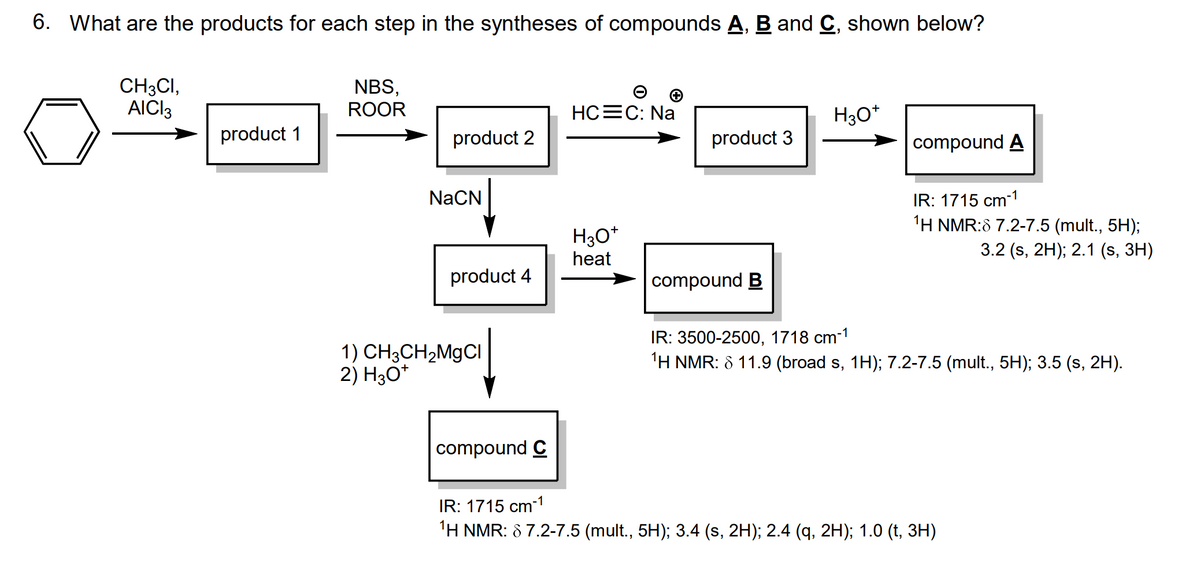
help needed with q 6. Thank you!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- A key step in the synthesis of naproxen, an NSAID more commonly known by its brand name, Aleve (Section 3.9), is a coupling reaction of 2-bromo-6-methoxynaphthalene to form 2-methoxy-6-vinylnaphthalene. Show three different coupling reactions, and the required reagents, that could be used tocarry out this step.Give the major organic product of each reaction of γ‑valerolactone with each of the given six reagents under the conditions indicated. Do not draw any byproducts formed. Reagent 1. NaOH, H2O, heat, then H+, H2O 2. (CH3)2CHCH2CH2OH (excess), H+ 3. (CH3CH2)2NH and heat 4. CH3MgI (excess), ether, then H+/H2O 5. LiAlH4, ether, then H+/H2O 6. DIBAL, toluene, low temperature, then H+/H2OA key step in the synthesis of naproxen, an NSAID more commonly known by its brand name, Aleve (Section 3.9), is a coupling reaction of 2-bromo6-methoxynaphthalene to form 2-methoxy-6-vinylnaphthalene. Show three different coupling reactions, and the required reagents, that could be used to carry out this step.
- Nitrating methyl benzoate. 1.Describe the mechanism of EAS clearly and in detail 2. What is the effect of subtituent of the outcome of EAS;include 3 classes of subtituentsDoxaprost, an orally active bronchodilator patterned after the natural prostaglandins , is synthesized in the following series of reactions starting with ethyl 2-oxocyclopentanecarboxylate. Except for the Nef reaction in Step 8, we have seen examples of all other types of reactions involved in this synthesis. Q. Step 11 can best be described as a Grignard reaction with methylmagnesium bromide under very carefully controlled conditions. In addition to the observed reaction, what other Grignard reactions might take place in Step 11?-Reactions E1 and E2 with AlcoholsIt develops the following reaction mechanisms and the main and secondary products:
- Determine the reagents A, B, C, D, E and the structure of compounds I, II, II in the reaction belowIn each of the following electrophilic substitution reactions (i) as a first step show the“preactivation” or generation of an electrophilic species; (ii) using arrows provide a mechanisticexplanation of the course of the reaction (show all resonance structures contributing for intermediatearenium ion stabilization); (iii) write all major products of the reaction. Friedel-Crafts acylation of anisole with 2-methylpropanoyl chloride ((CH3)2-CH-COCl) inthe presence of AlCl3 as a catalyst.3-Bromocyclohexene is a secondary halide, and benzyl bromide is a primary halide. Both halides undergo SN1 substitution about as fast as most tertiary halides. Use resonance structures to explain this enhanced reactivity.
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?

