Yes. H No, it has the wrong number of valence electrons. The correct number is: 0 Н—о— Н No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. :H – The correct number is: U No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. :0-c-0: The correct number is: O No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* O * If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many times as necessary, For example, if two oxygen atoms don't satisfy the octet rule, enter " .0".
Yes. H No, it has the wrong number of valence electrons. The correct number is: 0 Н—о— Н No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. :H – The correct number is: U No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* Yes. No, it has the wrong number of valence electrons. :0-c-0: The correct number is: O No, it has the right number of valence electrons but doesn't satisfy the octet rule. The symbols of the problem atoms are:* O * If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many times as necessary, For example, if two oxygen atoms don't satisfy the octet rule, enter " .0".
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter3: Atomic Shells And Classical Models Of Chemical Bonding
Section: Chapter Questions
Problem 94AP: The molecular ion S3N3 has the cyclic structure All SN bonds are equivalent. (a) Give six...
Related questions
Question
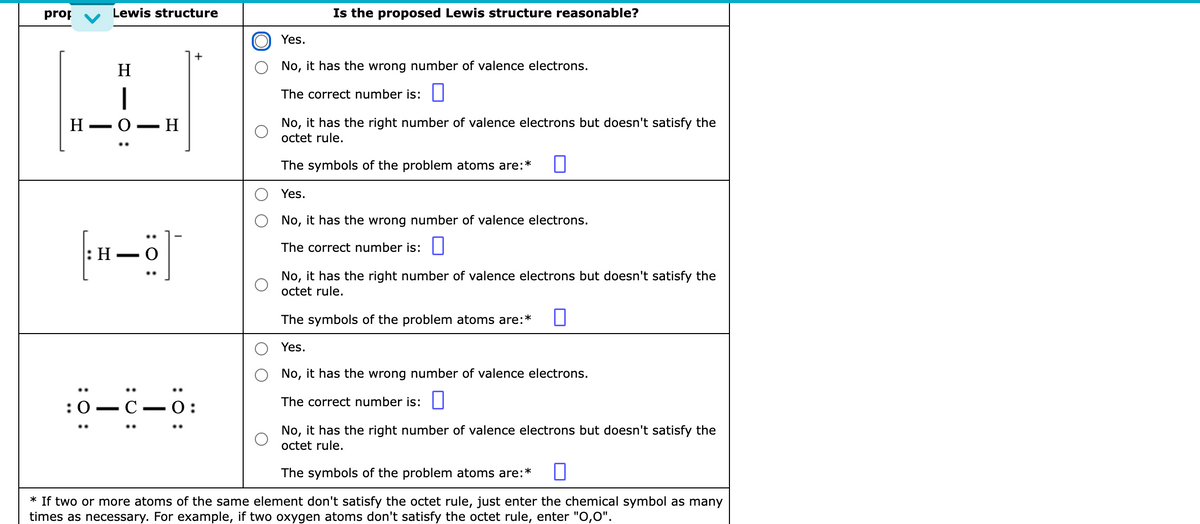
Transcribed Image Text:prop
Lewis structure
Is the proposed Lewis structure reasonable?
Yes.
H
No, it has the wrong number of valence electrons.
|
The correct number is: ||
Н— О — Н
No, it has the right number of valence electrons but doesn't satisfy the
octet rule.
The symbols of the problem atoms are:*
Yes.
No, it has the wrong number of valence electrons.
The correct number is: ||
|:Н — О
No, it has the right number of valence electrons but doesn't satisfy the
octet rule.
The symbols of the problem atoms are:*
Yes.
No, it has the wrong number of valence electrons.
..
:0 — С —О:
The correct number is: ||
No, it has the right number of valence electrons but doesn't satisfy the
octet rule.
The symbols of the problem atoms are:*
* If two or more atoms of the same element don't satisfy the octet rule, just enter the chemical symbol as many
times as necessary. For example, if two oxygen atoms don't satisfy the octet rule, enter "O,0".
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning