
Concept explainers
Which block in each of the following drawings of a balance is more dense, red or green? Explain.
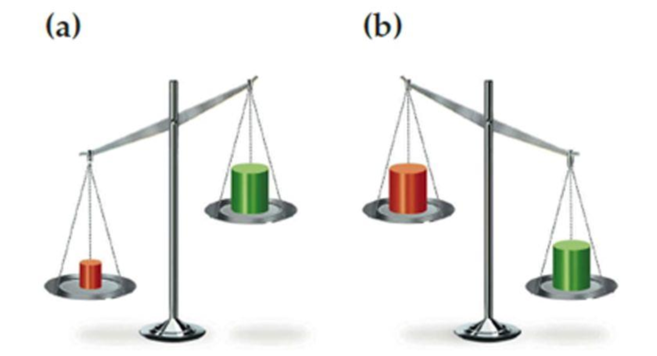
a)
Interpretation:
The denser block in drawing of a balance has to be identified and explained.
Concept Introduction:
Density:
Density can be defined as property that links mass of an object to its volume. It derived quantity and its unit is
Explanation of Solution
It is known that,
Density is directly proportional to mass and inversely proportional to volume.
In (a), red is identified as denser block because it is found have more mass and less volume than green block.
b)
Interpretation:
The denser block in drawing of a balance has to be identified and explained.
Concept Introduction:
Density:
Density can be defined as property that links mass of an object to its volume. It derived quantity and its unit is
Explanation of Solution
It is known that,
Density is directly proportional to mass and inversely proportional to volume.
In (b), green is identified as denser block because it is found have more mass and less volume than green block.
Want to see more full solutions like this?
Chapter 0 Solutions
General Chemistry: Atoms First
- Consider each of the following balances. Which one is the most precise? Which one is the least? How much uncertainty is associated with each balance?arrow_forwardReferring to Table 2.8, determine whether magnesium, ethanol, silver, or salt is the least dense.arrow_forwardReferring to Table 2.8, which substance listed is most dense? Which substance is least dense? For the two substances you have identified, for which one would a 1.00-g sample occupy the larger volume?arrow_forward
- Rank the substances in the photograph from least dense to most dense. Explain your reasoning.arrow_forwardState the volume of liquid in each graduated cylinder in the figure below and explain how you decided upon the appropriate number of significant figures. The markings are calibrated to be read at the bottom of the curved surface, and the numbers represent milliliters.arrow_forwardWithout doing any calculations, determine whether substance A with a density of 1.0g/cm3 is more or less dense than substance B with a density of 1.0kg/m3. a.substanceAb.substanceBarrow_forward
- A student checked the accuracy of two standard top-loading balances by testing them with a standard 5.000-g mass. The results were as follows: Balance 1: 4.99 g, 5.04 g, 5.03 g, 5.01 g Balance 2: 4.97 g, 4.99 g, 4.95 g, 4.96 g Calculate the average values for balances 1 and 2 and calculate the percent error for each. Which balance is more accurate?arrow_forwardFor each of the following figures, a through d, decide which block is more dense: the orange block, the blue block, or it cannot be determined. Explain your answers.arrow_forwardCompare and contrast the multiplication/division significant figure rule to the significant figure rule applied for addition/subtraction in mathematical operations.arrow_forward
- A 38.6 kg marble slab is shown above what is its density give your answer to the nearest tenth _ g/cm3arrow_forwardBalance A: 15.746 gBalance B: 15.75 gBalance C: 15.7 g When the liquid was added to the beaker, a buret was used to deliver 20.10 mL. Given thatdensity = mass/volume, calculate the density of the unknown liquid using the mass resultsfrom each balance along with this volume. Make sure you report each result to the correctnumber of significant figures. Balance A: g/mLBalance B: g/mLBalance C: g/mLarrow_forwardWhy is that Fahrenheit need to convert to Celsius? Is it easy to do a mathematical method if we use Celsius in solving?arrow_forward

 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





