
Concept explainers
(a)
Interpretation:
The place of alkali metals in the periodic table has to be indicated
(a)
Explanation of Solution
The alkali metals are indicated in pink color in the periodic table.
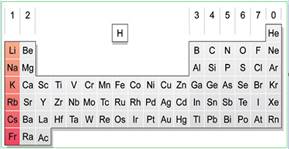
Figure-1
The first group metals are called as alkali metals. Generally it’s more reactive in nature.
(b)
Interpretation:
The place of halogens in the periodic table has to be indicated
(b)
Explanation of Solution
The Halogens are indicated in red color in the periodic table.

Figure-2
The
(c)
Interpretation:
The place of alkaline metals in the periodic table has to be indicated
(c)
Explanation of Solution
The alkaline earth metals are indicated in green color in the periodic table.
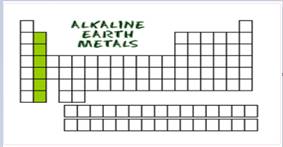
Figure-3
The second group metals are called as alkaline earth metals. Generally it’s second most reactive in nature.
(d)
Interpretation:
The place of
(d)
Explanation of Solution
The transition elements can be indicated using pink color in the periodic table.
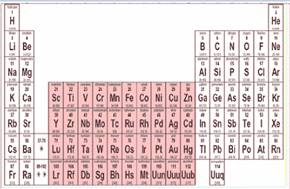
Figure-4
The
(e)
Interpretation:
The place of Hydrogen in the periodic table has to be indicated
(e)
Explanation of Solution
The Hydrogen atom is indicated in red color in the periodic table.
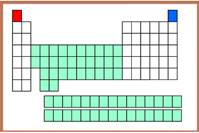
Figure-5
The first group and first element called as Hydrogen atom in the periodic table.
(f)
Interpretation:
The place of helium in the periodic table has to be indicated
(f)
Explanation of Solution
The Helium element can be indicated using blue color in the periodic table.
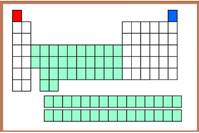
Figure-6
The
Want to see more full solutions like this?
Chapter 1 Solutions
General Chemistry: Atoms First -Laboratory Manual
- Which of the following are isotopes of element X, the atomic number for which is 9: 919X, 920X, 189X, and 921X?arrow_forward2.46 Why are nonmetals important even though they account for only a very small fraction of the elements in the periodic table?arrow_forwardThe following table presents the abundances and masses of the isotopes of zinc. What is the atomic weight of zinc?arrow_forward
- What is the name of the element in Group 4A and Period 5?arrow_forwardGiven that the periodic table is an organizational scheme for the elements, what might be some other logical ways in which to group the elements that would provide meaningful chemical information in a periodic table of your own devising?arrow_forward2-69 (Chemical Connections 2A) Why does the body need sulfur, calcium, and iron?arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





