
Organic Chemistry
2nd Edition
ISBN: 9781111807955
Author: Wyatt
Publisher: Cengage
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 13CTQ
Use VSEPR to assign a value of (close to) 109.5, 180 or 120 to each marked bond angle.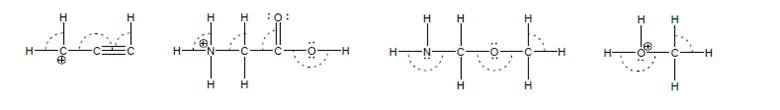
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the following bonds in order of increasing stretching frequency (cm-1) in IRspectroscopy:
Draw a bond-line structure that best matches the given 3D representation?
Homolytic bond breaking happens when there is an equal separation of electron in the presence of UV. true or false?
Chapter 1 Solutions
Organic Chemistry
Ch. 1 - (E) What does the number (+Z) at the center of...Ch. 1 - Prob. 2CTQCh. 1 - Prob. 3CTQCh. 1 - Prob. 4CTQCh. 1 - Prob. 5CTQCh. 1 - Prob. 6CTQCh. 1 - Prob. 7CTQCh. 1 - You hear a student from a nearby group say that...Ch. 1 - Use VSEPR to explain why the HBH bond angle of BH3...Ch. 1 - Both the HCH and HCO bond angles of H2CO...
Ch. 1 - Prob. 11CTQCh. 1 - Consider the following flat drawing of methane...Ch. 1 - Use VSEPR to assign a value of (close to) 109.5,...Ch. 1 - A student draws the picture of ammonia (NH3) in...Ch. 1 - Prob. 15CTQCh. 1 - How many central atoms does the molecule H2NCH3...Ch. 1 - Indicate the bond angle and shape about each...Ch. 1 - Explain how there can be two kinds of bent:...Ch. 1 - A student makes the following statement: “The...Ch. 1 - A student who missed this class needs to know how...Ch. 1 - Prob. 1ECh. 1 - Prob. 2ECh. 1 - Consider the incomplete valence shell...Ch. 1 - How many valence electrons does a neutral a. K...Ch. 1 - Consider the molecules AlCl3 (aluminum chloride)...Ch. 1 - Draw an example of a bent molecule with a bond...Ch. 1 - Label each atom marked with an arrow with the...Ch. 1 - a model of each of the following molecules: a....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Both the HCH and HCO bond angles of H2CO (formaldehyde) are very close to 120°, but oneis slightly smaller than the other. Predict which is smaller, and explain your reasoning.arrow_forwardWhich of the ff pairs of structure represent resonancr forms, and which do not?arrow_forwardCan you please see if the line bond structures are correctly drawn and then help with part b and c (i.e. numbering the peaks that would be seen and labeling the diasterotopic and enantiotropic atoms/groupsarrow_forward
- What happens to the bond angle (column 4) as the # of lone pairs (column 2) increases? Based on your answer above, do lone pairs or bonds take up more space?arrow_forwardThe LiH dipole torque is 1.964 * 〖10〗^(-30) Cm and the bond length is 1.596 A. Find the percentage of ionic Percent purity.arrow_forwardPolarity (a 3-d representation with bond and molecular dipoles annotated) of ClO4- , PF3, H2O, PF5 , SF4arrow_forward
- Polarity (a 3-d representation with bond and molecular dipoles annotated) of HCN, BeF2, SO2, O3, OF2, BF3arrow_forwardConstruct the CH3Br molecule in MolView and select the most accurate value of the H-C-H bond angle below.arrow_forwardHow many equivalent hydrogens are in CH2BrCl? (i.e., do they have identical environments with respect to the other atoms adjacent to themselves). A) 0 B) 3 C) 1 D) 2arrow_forward
- Give a clear handwritten answer with explanation please give answer all sub parts..give the geometry given below molecules ..a) dimine(N2H2) b) hydrogen peroxide c)NO2 d)radical HO2arrow_forwardProvide the Lewis, structure, Molecule/Ion type (i.e. AX3E, AX5, etc), Molecular Geometry, bond angle (the ideal/pure bond angle, not the bond angle as affect by lone pairs), and Polarity (a 3-d representation with bond and molecular dipoles annotated) "Exclude IF5"arrow_forwardDraw Frost circles for cyclobutadiene and benzene. Label all bonding, non bonding, and anti bonding orbitals. Which is more stable? Why?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY