
Radioactive wastes are packed in a long, thin-walledcylindrical container. The wastes generate thermal energynonuniformly according to the relation
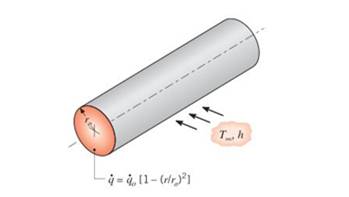
Obtain an expression for the total rate at which energyis generated in a unit length of the container. Use this result to obtain an expression the temperature
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Fundamentals of Heat and Mass Transfer
- Initially 100 milligrams of a radioactive substance was present. After 6 hours the mass had decreased by 5%. If the rate of decay is proportional to the amount of the substance present at time t, find the amount remaining after 24 hours. Determine the half life of the radioactive substance.arrow_forwardthermodynamics. The dominant force of attraction for CO2 whose density is 1.98 gram per liter at 273 kelvin temperature and 10 atmospheric pressure condition are ________ forces.arrow_forwardA balloon filled with ideal Helium gas (a monatomic gas) is tethered outside, and over time the balloon contracts from a volume of 1.15×10^3 m^3 to a volume of 1.05×10^3 m^3 . Assume that as it is contracting, the balloon maintains a constant pressure of 1.00 atm ( 1.013×10^5 Pa). The balloon contains 50,000 moles of Helium gas. a) Complete the following: i. Sketch a PV diagram for this process below. Label the pressures and volumes relevant at the initial and final times. ii. Find the work done by the environment on the gas during this process. Show your work! iii. Answer the following question: Did energy in the form of work enter or leave the system of Helium gas during the contraction? b) Find the change in the internal energy of the Helium gas during the process. c) Find the amount of heat transfer to or from the environment during this process and then answer the following question: Was the environment (outside atmosphere) at a higher or lower temperature than the helium gas…arrow_forward
- Krypton in a closed system is compressed adiabatically from 94 K and 1 bar to a final pressure of 24 bar. Compute the required work. Assume krypton is an ideal gas. From Appendix B in the text, we can assume the heat capacity of krypton is independent of temperature and CP=2.5R , where R is the molar gas constant R=8.314 J/(mol K). For an ideal gas, recall CV=CP−R=1.5R. Report your answer in units of kJ/mol using three decimal places.arrow_forwardThe Helmholtz free energy of a liquid column that rises, due to surface tension, inside a capillary tube (Figure 1), as a function of the height, h, is given by: F(h) =(ro)gr^2h^2/2 = -2 pi sigma r h cos teta, where r is the radius of the tube, g is the local ac- celaration of gravity, is the surface tension of the liquid, and is the contact angle of the liquid in contact with the wall of the tube. (a) From this expression, obtain the value of h as a function of the other physical parameters at equilibrium.Calculate the height that water will rise in a capil-lary of diameter 0.05mm. Assume that the contactangle between the water and the tube is zero. Thesurface tension of water at experimental conditionsisσ= 7.73×10−2N/m, and the local accelarationof gravity isg= 9.7m/s2.arrow_forwardthe title of the book: Engineering Thermofluids Thermodynamics, Fluid Mechanics, and Heat Transfer by: Mahmoud Massoudarrow_forward
- A metallic container of fixed volume of 2.5 × 10−3 m3 immersed in a large tank of temperature 27 °C contains two compartments separated by a freely movable wall. Initially, the wall is kept in place by a stopper so that there are 0.02 mol of the nitrogen gas on one side and 0.03 mol of the oxygen gas on the other side, each occupying half the volume. When the stopper is removed, the wall moves and comes to a final position. The movement of the wall is controlled so that the wall moves in infinitesimal quasi-static steps. (a) Find the final volumes of the two sides assuming the ideal gas behavior for the two gases. (b) How much work does each gas do on the other? (c) What is the change in the internal energy of each gas? (d) Find the amount of heat that enters or leaves each gas.arrow_forward1. (SI Unit) If the latent heat of boiling water at 1.013 bar is 2257 kJ/kg, the total quantity of heat (kJ) which must be added to 1 kg of water at 30°C in order to boil it is: (20 points) *arrow_forwardPlease useBook: Introduction to Chemical Engineering Thermodynamics 8 edition, 2018 (Smith, J.M., Van Ness, H.C., Abbot, M.M., Swihart.)arrow_forward
- a) In a simple experiment, a solid with a diameter 3 mm and density 1500 kg/m3is released from a tall tower containing air at 25 oC and 1 atm. The inside diameter of the column is 0.15 m. The density of the air is assumed to be 1.2 kg/m3. List all your assumptions and estimate the minimum height of the tower to make sure the particle reaches its terminal velocity before it hits the bottom of the tower. b) In another experiment, 5 kg of the solid particles are to be fluidized with air at 1 atm and 25 oC. The diameter and density of the particles are 3 mm and 95 kg/m3, respectively.Using the Ergun Equation, determine whether the flow of the air is laminar orturbulent. List all your assumptions.arrow_forwardKrypton in a closed system is compressed adiabatically from 74 K and 1 bar to a final pressure of 24 bar. What is the final temperature in K? Assume krypton is an ideal gas. From Appendix B in the text, we can assume the heat capacity of krypton is independent of temperature and CP=2.5R , where R is the molar gas constant R=8.314 J/(mol K). For an ideal gas, recall CV=CP−R=1.5R. Report your answer in units of K using three decimal places.arrow_forwardA 5-feet diameter Chimney is designed to handle a flue gas produced in a steam power plant at a rate of 17 lb/s. The barometric pressure is 29.92-inch Hg. Outside air enters the combustion chamber at 90 F. The average temperature of the flue gas inside the Chimney is 572 F and its molecular weight is 30. Cv = 0.35, Rair = 53.342 ft-lb/lb-R. Calculate: 5. The total draft, feet of air. 6. The height of the Chimney, feetarrow_forward
 Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
Principles of Heat Transfer (Activate Learning wi...Mechanical EngineeringISBN:9781305387102Author:Kreith, Frank; Manglik, Raj M.Publisher:Cengage Learning
