
Concept explainers
(a)
Interpretation: The condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contain both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is shown as,
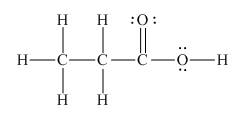
Figure 1
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula, one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 1
The Lewis structure of the given molecule is shown in Figure 1.
(b)
Interpretation: The condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is shown as, is,
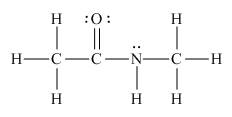
Figure 2
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula, one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 2
The Lewis structure of the given molecule is shown in Figure 2.
(c)
Interpretation: The condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is shown as,
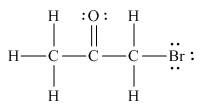
Figure 3
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula, one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 3
The Lewis structure of the given molecule is shown in Figure 3.
(d)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is,
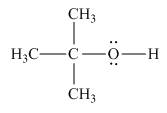
Figure 4
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 4
The Lewis structure of the given molecule is shown in Figure 4.
(e)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is,
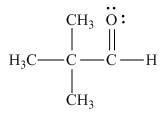
Figure 5
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 5
The Lewis structure of the given molecule is shown in Figure 5.
(f)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The the Lewis structure of the given molecule is,
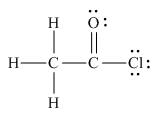
Figure 6
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three
oxygen and chlorine is bonded to carbon atom. Thus, the Lewis structure of the given molecule is,

Figure 6
The Lewis structure of the given molecule is shown in Figure 6.
(g)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is,
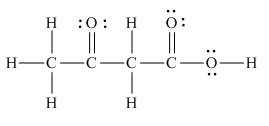
Figure 7
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 7
The Lewis structure of the given molecule is shown in Figure 7.
(h)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is,
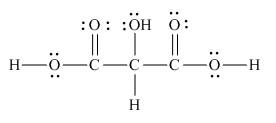
Figure 8
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 8
The Lewis structure of the given molecule is shown in Figure 8.
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry - With Access (Custom)
- Sketch the Lewis structures for CIF2+ and CIF2. How many lone pairs and bond pairs surround the Cl atom in each ion?arrow_forward1) Discuss the advantages and disadvantages of using the molecular models (ball and stick) as learning aids. Recommend an alternative to using this method for modeling molecules. 2) Build C6H6 as a ring. What do you notice about the placement of the bonds? This is an example of resonance. What is a resonant Lewis structure? 3) Explain how a diminished and expanded octet can be formed.arrow_forwardDraw a valid Lewis structure by using example: CH3CHO ?arrow_forward
- Write the Lewis symbols for Ca2+ and Cl-arrow_forwardName for Atl3 and name for KrBr2 with Lewis structure for KrBr2 also name for Tel6 and Lewis structure do all pleasearrow_forwardCompounds (1) C4H6Cl2 and (2) AlCl3. Explain the difference between them using the following categories. Types of bonding? The number of chlorine atom of each one?arrow_forward
- How many lone pairs are on each of the following atoms? (a) Br in HBr? (b) S in H2S? (c) C in CH4?arrow_forwardLewis structure for sulfur dioxide S02 Number of electrons set(groups)? Number of bonding electron sets (groups)? Number of non-bonding electron sets (groups) or lone pairs? How many bonds have a dipole ? If so do the dipole cancel each out?arrow_forwardA. The Lewis diagram for POCl3 is: The electron-pair geometry around the P atom in POCl3 is ______ There are _____ lone pair(s) around the central atom, so the geometry of POCl3 is ______ B. The Lewis diagram for BH2- is: The electron-pair geometry around the B atom in BH2- is ______ There are _____ lone pair(s) around the central atom, so the geometry of BH2- is _____ (c) The amount of acrylamide found in potato chips is 1.7 mg/kg. If a serving of potato chips is 35 g, how many moles of acrylamide are you consuming? ________molarrow_forward
- In the Lewis model, we use resonance forms to represent the two equivalent bonds. True or false? Explain how?arrow_forwardConsider the SO 3 molecule. How many valence electrons does the molecule have? How many lone pairs of electrons are on the central atom? How many lone pairs of electrons in total are on the substituent atoms? How many resonance structures can be drawn for the molecule? If the ion doesn't exhibit resonance, indicate "1" as only one structure can be drawn for the molecule.arrow_forwardDraw the Lewis Structure of CH3OCH3 Molecular Geometry? Ideal Bond Angles? Are there Polar Bonds Present (Indicate on Lewis do structure Is there an Overall Dipole? Octet rule violator and how?arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


