
Concept explainers
Ozone
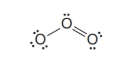
(a) Assign any necessary formal charges to the atoms in this structure. (b) Write another equivalent resonance structure for ozone. (c) What do these resonance structures predict about the relative lengths of the two oxygen-oxygen bonds of ozone? (d) In the structure above, and the one you have written, assume an angular shape for the ozone molecule. Is this shape consistent with VSEPR theory? Explain your answer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Living By Chemistry: First Edition Textbook
Chemistry For Changing Times (14th Edition)
Basic Chemistry (5th Edition)
Essential Organic Chemistry (3rd Edition)
Chemistry: Structure and Properties (2nd Edition)
Elementary Principles of Chemical Processes, Binder Ready Version
- Draw the three resonance structures for (a) the cyanate ion (NCO−) and (b) the isocyanate ion (CNO−). In each case, rank the resonance structures in order of increasing importance. Be sure to include all lone pair electrons and nonzero formal charges.a) NCO−arrow_forwardWrite the correct Lewis structure and assign a formal charge to each atom in fulminate ion, CNO.arrow_forwardCarbon monoxide (CO) is an example of an overall neutral molecule (netcharge=0) that hasnon-zero formal charges. Draw a Lewis structure of carbon monoxide (CO).arrow_forward
- Formamide, HC(O)NH2, is prepared at high pressures from carbon monoxide and ammonia, and serves as an industrial solvent (the parentheses around the O indicate that it is bonded only to the carbon atom and that the carbon atom is also bonded to the H and the N atoms). Two resonance forms (one with formal charges) can be written for formamide. Write both resonance structures, and predict the bond angles about the carbon and nitrogen atoms for each resonance form. Are they the same? Describe how the experimental determination of the HNH bond angle could be used to indicate which resonance form is more important.arrow_forwardWrite the two resonance structures for the pyridinium ion, C5H5NH+arrow_forwardThe following is a skeleton of a molecular anion having the overall formula C7H6NO–. The H atoms are not shown. Starting with the structure drawn below, complete the Lewis structure by adding all H atoms, valence electrons, and π bonds. This structure will have a –1 formal charge on the oxygen.arrow_forward
- Write the Lewis structures of both isomers with the formula C2H7N.arrow_forwardAssuming acid strength relates directly to electronegativityof the central atom, rank H₃PO₄, HNO₃, and H₃AsO₄ in order of increasing acid strength.arrow_forwardDraw two electron-dot resonance structures that obey the octet rule for trichloronitromethane, CCl3NO2, and show the formal charges on N and O in both structures. (Carbon is connected to the chlorines and to nitrogen; nitrogen is also connected to both oxygens.)arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


