
Concept explainers
(a)
Interpretation:
The type of intermolecular force present in Chloroform has to be given.
Concept introduction:
Intermolecular forces:
Intermolecular forces are like cohesive forces, acting between the molecules. The overall stability of a compound depends on how strong the molecules are held together. Intermolecular force is concerned about the overall stability of a substance. A stable substance has stronger intermolecular forces.
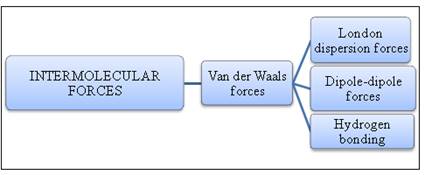
Figure 1
(b)
Interpretation:
The type of intermolecular force present in Oxygen has to be given.
Concept introduction:
Intermolecular forces:
Intermolecular forces are like cohesive forces, acting between the molecules. The overall stability of a compound depends on how strong the molecules are held together. Intermolecular force is concerned about the overall stability of a substance. A stable substance has stronger intermolecular forces.
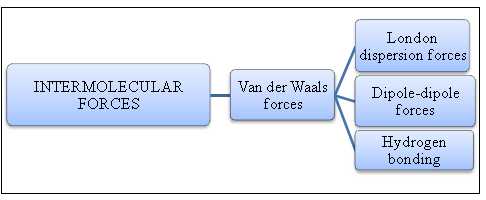
Figure 1
(c)
Interpretation:
The type of intermolecular force present in Polyethylene has to be given.
Concept introduction:
Intermolecular forces:
Intermolecular forces are like cohesive forces, acting between the molecules. The overall stability of a compound depends on how strong the molecules are held together. Intermolecular force is concerned about the overall stability of a substance. A stable substance has stronger intermolecular forces.

Figure 1
(d)
Interpretation:
The type of intermolecular force present in Methanol has to be given.
Concept introduction:
Intermolecular forces:
Intermolecular forces are like cohesive forces, acting between the molecules. The overall stability of a compound depends on how strong the molecules are held together. Intermolecular force is concerned about the overall stability of a substance. A stable substance has stronger intermolecular forces.

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
General Chemistry: Atoms First
- Potassium chloride has the same unit cell as NaCl. Using the ion sizes in Figure 7.11, calculate the density of KCl. FIGURE 7.11 Relative sizes of some common ions. Rodii are given in picometers (1 pm 1 1012 m). (Data taken from J. Emsley, The Elements, Clarendon Press, Oxford, 1998, 3rd edition.)arrow_forwardExplain why liquids assume the shape of any container into which they are poured, whereas solids are rigid and retain their shape.arrow_forwardIn groups and geometry, what kind of lattice do you obtain by v = (1, 0) and w = (2,-4)?arrow_forward
- #7# Polar or non polar ? For CF2CL2arrow_forwardWhich of the following compounds can exhibit hydrogen bonding? Explain why. NH3 (ammonia), H2O (water), HCl (used to make hydrochloric acid), or C4H10 (butane, used as fuel for cigarette lighters and portable stoves)arrow_forwardSketch the shape and orientation of the following types oforbitals: (a) s, (b) Pz?arrow_forward
- Calcium forms a face-centered cubic crystalline structure with a density of 1.55 g/cm3. What is its atomic radius (in pm)?arrow_forwardWhich intermolecular forces will be present in the substance SF4?arrow_forwardWhat kind of intermolecular force (s) are there between the following pairs of particles? NH3 and c6H6arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning



