
Interpretation:
The radius of Tungsten in picometers has to be calculated.
Concept Introduction:
Body-centered unit cell:
A body – centered cubic cell is a type of unit cell with atoms arranged in all the vertices of the unit cell and one atom at the center of the cube.
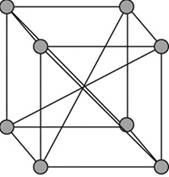
Figure 1
A body – centered cubic unit cell has its all the eight vertices arranged with atoms in it. Eight atoms are arranged in a simple cubic unit cell in eight vertices. Each atom in a vertex is shared by eight neighboring unit cells such that each cubic unit cell has
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
General Chemistry: Atoms First
- Why are capillary rises and depressions not seen for cylinders with large radii?arrow_forwardIn terms of the kinetic molecular theory, in what ways are liquids similar to solids? In what ways are liquids different from solids?arrow_forwardWhat are the two ways in which the escape of high-energy molecules from the surface of a liquid during the process of evaporation affects the liquid?arrow_forward
- Satellites in space often suffer from vacuum welding, in which two metal parts in contact tend to stick together more than expected over a period of time. Why does this phenomenon occur in space and not on Earth?arrow_forwardExplain why liquids assume the shape of any container into which they are poured, whereas solids are rigid and retain their shape.arrow_forwardRubidium chloride has the sodium chloride structure at normal pressures but assumes the cesium chloride structure at high pressures. (See Exercise 69.) What ratio of densities is expected for these two forms? Does this change in structure make sense on the basis of simple models? The ionic radius is 148 pm for Rb+ and 181 pm for CI.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





