
Concept explainers
Interpretation:
Calculate the height of water in meters that can be supported by standard atmospheric pressure and 1 bar if the density of water is 1.00g/mL and the density of mercury is 13.6 g/mL.
Concept introduction:
Pressure is force per unit area.
Its unit is Newton per meter square or we can say kilogram per meter per Second Square. These two are not the only units of pressure. The International system of unit (S.I unit) of pressure is Pascal, but these are not the only units of pressure. There are more units of pressure like pounds per square inch, atmospheric (atm), mm Hg, torr etc.
Pressure is measured in the U-tube as shown below: The pressure is measured by filling water into this tube so that the difference between the lengths of two sides of U-tube gives an idea about the pressure. In this tube, one end is kept at room atmospheric pressure and the other side to that whose pressure has to be measured. So, this is the reason for the different units of the pressure because the length can be measured in different units. In case of water, there would be a requirement of a large U-tube so, mercury is used instead of water because its density is more than that of water. So, another unit of pressure was invented that is mmHg.
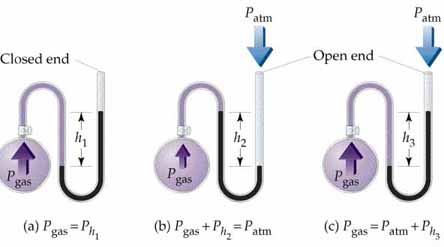
To determine:
The height of water in the open end manometer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Chemistry, Books a la Carte Edition and Modified Mastering Chemistry with Pearson eText & ValuePack Access Card (7th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





