
Concept explainers
(a)
Interpretation:
The given Lewis structure needs to be completed.
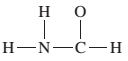
Concept Introduction:
The Lewis dot structure is the representation of a molecule or compound in which atoms are arranged or bonded in such a way that all the atoms have complete octets.
The bond formation between the atoms takes place due to the sharing of valence electrons between them while the remaining outer electrons are denoted as lone pair of electrons.
(b)
Interpretation:
The given Lewis structure needs to be completed.
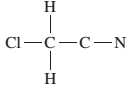
Concept Introduction:
The Lewis dot structure is the representation of a molecule or compound in which atoms are arranged or bonded in such a way that all the atoms have complete octets.
The bond formation between the atoms takes place due to the sharing of valence electrons between them while the remaining outer electrons are denoted as lone pair of electrons.
(c)
Interpretation:
The given Lewis structure needs to be completed.
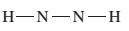
Concept Introduction:
The Lewis dot structure is the representation of a molecule or compound in which atoms are arranged or bonded in such a way that all the atoms have complete octets.
The bond formation between the atoms takes place due to the sharing of valence electrons between them while the remaining outer electrons are denoted as lone pair of electrons.
(d)
Interpretation:
The given Lewis structure needs to be completed.
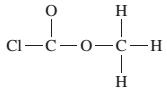
Concept Introduction:
The Lewis dot structure is the representation of a molecule or compound in which atoms are arranged or bonded in such a way that all the atoms have complete octets.
The bond formation between the atoms takes place due to the sharing of valence electrons between them while the remaining outer electrons are denoted as lone pair of electrons.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
EBK BASIC CHEMISTRY
- does it compare with the known melting and boiling point? (1) 9. The average kinetic energy of water molecules is a measure of the temperature of water. When the temperature of water remains constant the average kinetic energy of the molecules remains constant, even though the water is being heated by the Bunsen flame. So, energy is being taken in by the water, but it is not being used to increase the kinetic energy of the molecules. 9.1 What type of energy are the water molecules gaining during a phase change? (1) 9.2 Explain your reasoning (to question 9.1) with reference to the kinetic theory of matter. 10. Write the conclusion. (3) [30]arrow_forwardYou have a large bottle of 10.0 M hydrochloric acid and you want to make 6.0 L of 3.0 M hydrochloric acid. How much concentrated do you need and how much water? (1.8, 4.2)arrow_forwardBarium Hydroxide has a solubility of 4.68 g Ba(OH)2 in 100. g H2O at 25°C. Determine if each of the following solutions will be saturated or unsaturated at 25°C: (9.3) a) adding 32 g of Ba(OH)2 to 990 g of H2O b) adding 7.0 g of Ba(OH)2 to 125 g of H2O c) adding 22 g of Ba(OH)2 to 350. g of H2Oarrow_forward
- (9.4) Which of the following elements has the most number of valence electrons? selenium O antimony helium O bariumarrow_forwardshould react most rapidly with Cl2 and AICI. (9.28.h) should have the highest boiling point, 7. Of the compounds shown below, B A. D. 8. Of the compounds shown below, NO2 HO, is not aromatic. (9.1 lg,i) 9. Of the compounds shown below, only - D Z-I Uarrow_forward9.116 Lithium chloride has a solubility of 55 g of LiCl in 100. g of H,O at 25 °C. Determine if each of the following mixtures forms an unsaturated or saturated solution at 25 °C: (9.3) a. adding 10 g of LiCl to 15 g of H;O b. adding 25 g of LİCI to 50. g of H,0 c. adding 75 g of LiCl to 150. g of H,O ne on nearrow_forward
- 9.2 An unknown salt is found to contain 30,6% of water. The formula mass for anhydrous salt (AS) is 100 amu. 9.2.1 Calculate the amount of water bonded with an unknown salt. 9.2.2 An anhydrous salt above consists of a metal with a molar mass of 40. (a) Write down the chemical symbol of this metal. (b) To which group does the metal belongs? 9.2.3 Write down the chemical formula of an anhydrous salt above.arrow_forward(5.8)Which of the following reactions will form a gaseous product? O H₂CO3(aq) + Pb(NO3)2(aq) O NaOH(aq) + HNO3(aq) O None of these O Na₂SO3(aq) + H₂SO4(aq) ◄ Previousarrow_forwardThis is possible because of the following reactions that take place rapidly as long as reactants are available: (11.3) Equation : 51-(aq) + IO-3(aq) + 6H+(aq) ----> 3I2(aq) +3H2O(I)arrow_forward
- Name each of the following molecular compounds: b. SF6 e. SO₂ a. CBr4 d. N₂O4 c. BrCl f. CS₂ Write the formula for each of the following molecular compounds: (6.5) a. carbon sulfide c. dihydrogen sulfide b. diphosphorus pentoxide d. sulfur dichloridearrow_forward9.2 An unknown salt is found to contain 30,6% of water. The formula mass for anhydrous salt (AS) is 100 amu. 9.2.1 Calculate the amount of water bonded with an unknown salt.arrow_forwardshould react most rapidly with Clz and AICI. (9.28 h) 8. Of the compounds shown below, NO2 HO. D. 9. Qarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





