
Interpretation:
The 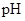 value,
value,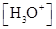 and
and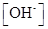 concentration for given weak acid should be determined.
concentration for given weak acid should be determined.
Concept Introduction:
Weak Acids: Acids that do not easily dissociate into ions completely which has difficulty in proton donation are considered as weak acids. Weak acid forms stronger conjugated base.
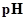 : The concentration of hydrogen ion is measured using
: The concentration of hydrogen ion is measured using 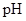 scale. The
scale. The 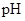 of a solution is a figure that expresses the acidity or the alkalinity of a given solution.
of a solution is a figure that expresses the acidity or the alkalinity of a given solution.
It is defined as the negative base-10 logarithm of the hydrogen or hydronium ion concentration.
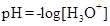
Ionic-product constant for water: It is the hydronium ion concentration times the 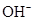 concentration present in the solution.
concentration present in the solution.
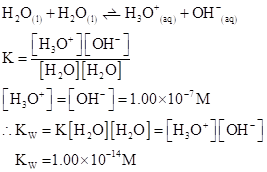
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
- What is the pH of a solution that has OH– concentration of 10-9 M?arrow_forwardIn order to make a solution of the concentration 0.364 M, what mass of NaCl should bedissolved in 152 mL of water?arrow_forwardA solution has a hydrogen ion concentration of 0.01 mol/L. What is its pH? What is its hydroxide ion concentration? Is it acidic, basic, or neutral? How does the hydrogen ion concentration of this solution differ from one with a pH of 1?arrow_forward
- A buffer solution contains an equal concentration of weak acid HX and its conjugate base ion X™. The ionization constant Ka of HX is 2.2 x 10-8. What is the pH of the buffer?arrow_forwardWhat is the pH of a solution of 100 ml of 0.01 M H3PO4 and 100 ml of 0.01 M Na3PO4?arrow_forwardIf 0.752 moles of (NH3OH)Cl is dissolved in 1 L of water what is the pH of the solution?arrow_forward
- A buffer solution is composed of 1.00 mol of acid and 1.75 mol of the conjugate base. If the pKa of the acid is 3.60, what is the pH of the buffer? pH=arrow_forwardA 0.200 M solution of a weak monoprotic acid (HA) has a pH of 2.35. What is the value of K of this acid? a K =2.011e-4 a What is the percent ionization of this acid? Percent Ionization = .10 %arrow_forwardfor 0.9% NaCl solution and 10% NaCl solution, which one has higher concentration of WATER?arrow_forward
- A 35 mL of solution of hydrochloric acid is neutralized by 15 mL of 0.5 M potassium hydroxide. What is the concentration of hydrochloric acid? The balanced equation is HCI + KOH - H,O + KCI O 0.75 M O 0.25 M O 0.214 M O 0.786 Marrow_forwardThe pHpH scale for acidity is defined by pH=−log10[H+] where [H+]is the concentration of hydrogen ions measured in moles per liter (M). A solution has a pH of 10.2. Calculate the concentration of hydrogen ions in moles per liter (M).arrow_forwardIf the pH of a voledronic acid solution is 5.8, and the voledronate concentration is 9 mM, what is the concentration of voledronic acid? (pKa=5.0) 1.4 0.5 185.2 20 379.5arrow_forward
 Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning
Biology (MindTap Course List)BiologyISBN:9781337392938Author:Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. BergPublisher:Cengage Learning


