
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
4th Edition
ISBN: 9780134465715
Author: John E. McMurry, David S. Ballantine
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 10.2, Problem 10.3CIAP
Interpretation Introduction
Interpretation:
The conjugate acid and base pairs present in given reaction should be identified and to which side the reaction is favored should be indicated.
Concept Introduction:
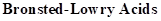 : If a species loses a proton then it is considered as
: If a species loses a proton then it is considered as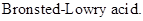
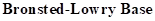 : If a species receives one proton, then it is considered as
: If a species receives one proton, then it is considered as 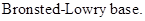
If a base receives one proton, then the formed species is a conjugate acid whereas an acid lose one proton, then the formed species is a conjugated base.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Given the titration curve of the hypothetical polyprotic acid X at 0.100 M concentration (pKa1=4.0, pKa2=8.0, pKa3=12.0) titrated with 0.600 M NaOH, identify the pH at point C, H, E, and M.
The ionization of p-nitrophenol is shown below (pKa = 7.0):
a. Identify the weak acid and conjugate base.
b. At pH 7, what are the relative concentrations of ionized and un-ionized p-nitrophenol?
c. If enough concentrated hydrochloric acid is added to a solution of p-nitrophenol to lower the pH from 7 to 5, what will happen to the relative concentrations of the ionized and un-ionized forms?
d. Ionized p-nitrophenol has a yellow color, while the un-ionized form is colorless. The yellow color can be measured using a spectrophotometer at 400nm. In order to determine the total amount of p-nitrophenol in a solution, would you perform the spectrophotometer reading at an acidic or basic pH? Clearly explain why?
e. A solution of p-nitrophenol at pH 7.95 was found to have an A400 of 0.255 . What is the total concentration (in µM) of p-nitrophenol (ionized plus un-ionized) in the solution? The molar extinction coefficient of p-nitrophenol is 18,500 M-1cm-1 and the pKa is 7.
Identify the conjugate acid-base pairs in the following reactions:
HNO2(aq) + H2O(l) → NO2 – (aq) + H3O+(aq)
_______ ______ _________ ________
CH3NH2 + H2O(l) → CH3NH3+ + OH –
_______ ________ ________ _________
Chapter 10 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
Ch. 10.1 - Which of the following are BrnstedLowry acids?...Ch. 10.1 - Prob. 10.2PCh. 10.1 - Prob. 10.3PCh. 10.1 - Prob. 10.4KCPCh. 10.2 - The concentration of HCl when released to the...Ch. 10.2 - Prob. 10.2CIAPCh. 10.2 - Prob. 10.3CIAPCh. 10.2 - Prob. 10.5PCh. 10.2 - Prob. 10.6PCh. 10.2 - Prob. 10.7P
Ch. 10.2 - Prob. 10.8PCh. 10.2 - Prob. 10.9KCPCh. 10.3 - Prob. 10.10PCh. 10.4 - Prob. 10.11PCh. 10.5 - Prob. 10.12PCh. 10.5 - Prob. 10.13PCh. 10.5 - Prob. 10.14PCh. 10.6 - Identify the following solutions as acidic or...Ch. 10.6 - Calculate the pH of the following solutions and...Ch. 10.6 - What is the pH of a 0.0025 M solution of HCl?Ch. 10.6 - Prob. 10.4CIAPCh. 10.6 - Prob. 10.5CIAPCh. 10.7 - How many equivalents are in the following? (a) 5.0...Ch. 10.7 - Prob. 10.19PCh. 10.8 - Maalox, an over-the-counter antacid, contains...Ch. 10.8 - Prob. 10.21PCh. 10.8 - Prob. 10.22PCh. 10.8 - Show how ethylamine (C2H5NH2) reacts with...Ch. 10.9 - Predict whether the following salts produce an...Ch. 10.10 - What is the pH of 1.00 L of the 0.100 M...Ch. 10.10 - Prob. 10.26PCh. 10.10 - Prob. 10.27PCh. 10.10 - A buffer solution is prepared using CN-(from NaCN...Ch. 10.11 - A titration is carried out to determine the...Ch. 10.11 - Prob. 10.30PCh. 10.11 - Prob. 10.31PCh. 10.11 - Prob. 10.32PCh. 10.11 - Prob. 10.6CIAPCh. 10.11 - Prob. 10.7CIAPCh. 10 - Prob. 10.33UKCCh. 10 - Prob. 10.34UKCCh. 10 - The following pictures represent aqueous acid...Ch. 10 - Prob. 10.36UKCCh. 10 - Prob. 10.37UKCCh. 10 - Prob. 10.38APCh. 10 - What happens when a weak acid such as CH3CO2H is...Ch. 10 - What happens when a strong base such as KOH solved...Ch. 10 - Prob. 10.41APCh. 10 - Prob. 10.42APCh. 10 - Prob. 10.43APCh. 10 - Prob. 10.44APCh. 10 - Prob. 10.45APCh. 10 - Prob. 10.46APCh. 10 - Label the BrnstedLowry acids and bases in the...Ch. 10 - Write the formulas of the conjugate acids of the...Ch. 10 - Write the formulas of the conjugate bases of the...Ch. 10 - Prob. 10.50APCh. 10 - Prob. 10.51APCh. 10 - Prob. 10.52APCh. 10 - Prob. 10.53APCh. 10 - Prob. 10.54APCh. 10 - Write the expressions for the acid dissociation...Ch. 10 - Based on the Ka values in Table 10.3, rank the...Ch. 10 - Prob. 10.57APCh. 10 - A 0.10 M solution of the deadly poison hydrogen...Ch. 10 - Prob. 10.59APCh. 10 - Prob. 10.60APCh. 10 - What is the approximate pH of a 0.02 M solution of...Ch. 10 - Calculate the pOH of each solution in Problems...Ch. 10 - Prob. 10.63APCh. 10 - What are the OH concentration and pOH for each...Ch. 10 - What are the H3O+ and OH concentrations of...Ch. 10 - Prob. 10.66APCh. 10 - Prob. 10.67APCh. 10 - Write balanced equations for proton-transfer...Ch. 10 - Sodium bicarbonate (NaHCO3), also known as baking...Ch. 10 - Refer to Section 10.8 to write balanced equations...Ch. 10 - Prob. 10.71APCh. 10 - For each of the following salts, indicate if the...Ch. 10 - Which salt solutions in problem 10.72 could be...Ch. 10 - Prob. 10.74APCh. 10 - Prob. 10.75APCh. 10 - Prob. 10.76APCh. 10 - Which of the following buffer systems would you...Ch. 10 - What is the pH of a buffer system that contains...Ch. 10 - Consider 1.00 L of the buffer system described in...Ch. 10 - Prob. 10.80APCh. 10 - Prob. 10.81APCh. 10 - Prob. 10.82APCh. 10 - How does normality compare to molarity for...Ch. 10 - Prob. 10.84APCh. 10 - Prob. 10.85APCh. 10 - Prob. 10.86APCh. 10 - Prob. 10.87APCh. 10 - Prob. 10.88APCh. 10 - Prob. 10.89APCh. 10 - Prob. 10.90APCh. 10 - Prob. 10.91APCh. 10 - Titration of a 12.0 mL solution of HCl requires...Ch. 10 - Prob. 10.93APCh. 10 - Titration of a 10.0 mL solution of NH3 requires...Ch. 10 - If 35.0 mL of a 0.100 N acid solution is needed to...Ch. 10 - For the titrations discussed in Problems 10.92 and...Ch. 10 - Prob. 10.97APCh. 10 - Prob. 10.98CPCh. 10 - Prob. 10.99CPCh. 10 - Prob. 10.100CPCh. 10 - Prob. 10.101CPCh. 10 - Prob. 10.102CPCh. 10 - Prob. 10.103CPCh. 10 - Prob. 10.104CPCh. 10 - Prob. 10.105CPCh. 10 - Prob. 10.106CPCh. 10 - Prob. 10.107CPCh. 10 - Prob. 10.108CPCh. 10 - Obtain a package of Alka-Seltzer, an antacid, from...Ch. 10 - Prob. 10.110GPCh. 10 - Prob. 10.111GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- One liter of a 0.1 M Tris buffer (pKa of Tris = 8.3, see Table 2.4) is prepared and adjusted to a pH of 2.0. A) What are the concentrations of the conjugate base and weak acid at this pH?Answer with 2 significant digits. I have solved for [HA] = 0.10 Can you please help solve for [A-] = ?arrow_forwardRefer to the following titration curve below: 13 12 11 10 9 7 6 5 4 3 4 6 8 10 12 14 16 18 20 22 24 26 28 30 Volume of Titrant / mL - Unknown Acid 0.10 mol/L - titrant = NaOH 0.1 mol/L At ph 10.0, which form of histidine is most abundant? His2+ His- His° His+arrow_forwardDetermine the net charge on the following pentapeptide "Lys-Ala-His-Asp-Ser" at: a. pH 1.0 b. pH 10.1arrow_forward
- Based on the following titration table of Glycine, would you be able to recreate the same for Histidine? Glicina Increasing pH H-N-CH,-COOH -H-N-CH,-co = H-N-CH,-C00- Net charge: +1 pH COOH-pKa = 2.3 NH3-pka = 9.6 CN AEq +1 +1 2.3 (0.5°-1)+(0.5°0)=-0.5 +0.5 0.5 9.6 -1 (0.5*+1)+(0.5°0)=+0.5 -0.5 1.5 14 Given that the pKas for Histidine are the following: pka1 = 1.82 pKa2 = 9.17 pКаз %3 6.00 pL = 7.59 2.arrow_forwardThe amino acid glycine is often used as an ingredient in buffers for biochemistry experiments. The amino group of glycine has a pKa of 9.6. Glycine exists in either a protonated form (-NH3+) or a free base (-NH₂). a) In what pH range can glycine be used as an effective buffer? b) In a 0.1M solution of glycine at pH 9.0, what fraction has its amino group in the protonated form? c) When 99% of the glycine is in the protonated from, what is the numerical relation between the pH of the solution and the pKa of the amino group?arrow_forwardA sample (660 mg) of a (homo)oligomeric protein of molecular weight of 132,000 Da was treated with an excess of 2,4-dinitro-fluorobenzene until the reaction was complete. The peptide bonds were then completely hydrolyzed by heating with 6 M hydrochloric acid. The hydrolysate was found to contain 5.5 mg of the following compound. NO2 O2N- NH НООС 2,4-Dinitrophenyl derivatives of the amino groups of other amino acids were not found. a). How many polypeptide chains are there in this protein? Show your work. b). Assuming that each of the subunit contains entirely a-helices, how many a-helical turns would there be, approximately, in each of the subunit? "For a protein of typical composition, average amino acid residue mass is ca. 110 Da."arrow_forward
- Draw dipeptide Arg-Thr at pH 6.0. Circle the six atoms that are restricted in one plane. Calculate its approximate pI assuming pKa for NH3+ is 10.0, pKa for COOH is 3.0, PKR for Arg is 12.0arrow_forwardGlutamic acid (1one of the 20 amino acids) has a side- chain carboxyl group (COOH, pKa = 4.3) as shown in Figures 4.7 and 4.8 a. Write the chemical equation for the dissociation of the side chain COOH. Label the weak acid and the conjugate base. b. The Henderson-Hasselbalch Equation (shown below) can be used to determine the ionization status of a weak acid: pH = pKa + log [base] Use the Henderson Hasselbalch equation to determine whether the glutamic acid side chain [acid] • carboxyl group is protonated or deprotonated at physiological pH.arrow_forwardAn enzyme (molecular weight= 24 kDa, pI= 5.5) is contaminated with two other proteins, one witha similar molecular mass and a pI of 7.0 while the other has a molecular mass of 100 kDa and a pIof 5.4. Suggest how the contaminated enzyme can be purified.arrow_forward
- The structure of chitosan, a polymer used to eliminate protein clumps that create wine haze in white wine, is shown below. The amine functional group on chitosan has a pka of 6.5 in its conjugate acid form. What is the charge state of the amine group of chitosan in wine with a pH of 3.6, given the following acid-base reaction, where chitosan is abbreviated as R? (If you need it, the Henderson-Hasselbach connection is also provided.) [RNH2] [RNH;] NH2 RNH2 + H20 2 RNH + 0H- pH = pKa+ log OH n a) At pH 3.6, it is negatively charged; b) At pH 3.6, it is positively charged; c) At pH 3.6, it is both positively and negatively charged; d) At pH 3.6, it is neutral.arrow_forwardAn enzyme uses a lysine and a histidine as general acid-base catalytic residues. In one direction of the reaction, the lysine and histidine residues are typically protonated (~NH3*, ~Im), but in the other direction of the reaction, the two residues are "reverse protonated" (~NH2, ~Hlm*) (see below scheme). A pH profile of the reaction at various pH values yielded estimates of 7.57 and 9.30 for the macroscopic ionization constants pk,' and pK2, respectively. A separate set of experiments with site-speciffic variants of the enzyme yielded an estimate of 8.78 for the microscopic ionization constant pKp. Given this information, what is the value of the microscopic ionization constant pKA? Report your answer to the nearest hundredth. Lys-NH3* His-HIm" KB H KA H* Lys-NH2 Lys-NH3* His-HIm" His-Im Kc H* Kp Lys-NH2 His-Imarrow_forward80mL of a 0.3M solution of hexapeptide Leu-His-Cys-Glu-Asn-Arg is adjusted to pH=pl. The solution is then titrated with 0.2M HCI to a final pH of 2.1. Sketch the titration curve, labelling the pH and volume axes. Indicate the volume of HCl needed to reach each relevant pKa value and equivalence point(s). Relevant pka values are: 2.1, 4.3, 6.0, 8.3, 9.8, and 12.5.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON