
Concept explainers
Use the following phase diagram for questions 9 and 10.
The following phase diagram is for salt water. There are four possible phases, which depend on the temperature and the sodium chloride content (NaCl).
- Ice and SC = Mixed ice and salt crystals.
- Ice and SW = Ice and salt water.
- SW = Salt water.
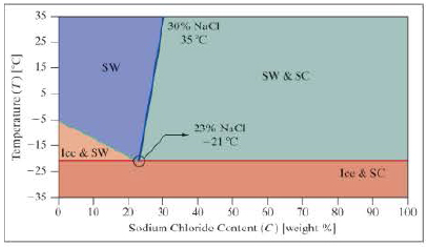
- SW and SC = Salt water and salt crystals.
There are often multiple ways to solve the same problem; here we look at a few alternative ways to determine the phase of the mixture.
- 9. a. In column C, develop the equation for the line dividing the phases of the ice–salt water mix and the salt water. Assume it was written in cell C11 and copied down.
- b. In column D, develop the equation for the line dividing the phases of the salt water and the salt water-salt crystals mix. Assume it was written in cell D11 and copied down.
- c. In column E, write an expression to determine the phase of the mixture.
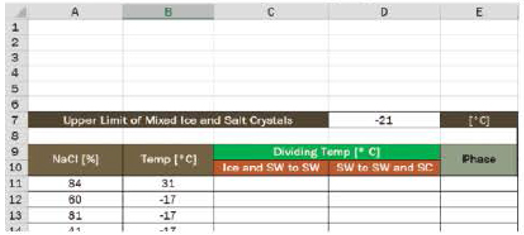
- d. Use conditional formatting to highlight the various phases. Provide a color key.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Thinking Like an Engineer: An Active Approach, Student Value Edition Plus MyLab Engineering with Pearson eText - Access Card Package (4th Edition)
Additional Engineering Textbook Solutions
Thermodynamics: An Engineering Approach
Heat and Mass Transfer: Fundamentals and Applications
Vector Mechanics for Engineers: Statics, 11th Edition
Vector Mechanics for Engineers: Statics and Dynamics
Vector Mechanics for Engineers: Statics and Dynamics
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
- Can someone please help me to solve question 10?Please show all work, formulas, and diagrams. Please and thank you!!!arrow_forwardHello, please help with this question for review purposes cuz my prof didn't teach us anything. Please provide charts and Table used with Plotting.arrow_forwardPLEASE SHOW SOLUION STEP BY STEP WITH UNITS. PLEASE ANSWER IT IN 30 MINS. TOPIC: THERMODYNAMICS - PROCESSES OF GASESarrow_forward
- Hello, I need help checking my answer, I got 0.064, is this correct?arrow_forwardThis question is the solution to the student’s number. My number is 39914140664014. The temperature starts at 39 degrees and ends with the last two numbers 14. This is for me.arrow_forwardCan someone please help me to solve question 8? Please show all work, formulas, and diagrams. Please and thank you!!!arrow_forward
- note: this is for engineering. Please answer ASAP! question: Four hundred grams of water at 20° C is placed inside a 100 grams glass container. Twenty grams of ice at 0° C is then added to the water. a. Draw the figure reflecting each substance quantities, process, change in temperature and objects of interest for the heat energy gain and heat energy loss b. Determine the temperature of the mixturearrow_forwardI need help matching the following terms to the phrase it describes. Answers are used only once.arrow_forwardPLEASE ANSWER COMPLETELY WITH BRIEF EXPLANATION EACH STEP. PROVIDE FBD'sarrow_forward
- There are a lot of subquestions in this question, but you can only answer a few of them, so I asked the exact same one twice, so please finish this one and move on to the next part, thank you!arrow_forwardPls help ASAP on this question. Pls do all of them. Just need one sentence for each question.arrow_forwardPlease show your solution and at the same time BOX and SPECIFY the answers that corresponds to each blank on the question. Make sure to fill all the blanks. Thank you. STRICKLY FOLLOW THIS: Calculated Answers Express your answers without rounding off and without scientific notation unless instructed otherwise. Multiple Blanks Write powers or subscript as is. Ex: Use b2 if you mean b2 or b2 Spell out Greek letters. Ex: Use pi if you mean greek letter pi Write answers without spaces. Ex: Use 2epsilon0r3 if you mean 3e0r3 Write fractions with a slash. Ex: Use 1/2arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





