
Prescott's Microbiology
11th Edition
ISBN: 9781260409062
Author: WILLEY, Joanne
Publisher: MCGRAW-HILL HIGHER EDUCATION
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 10.4, Problem 1MI
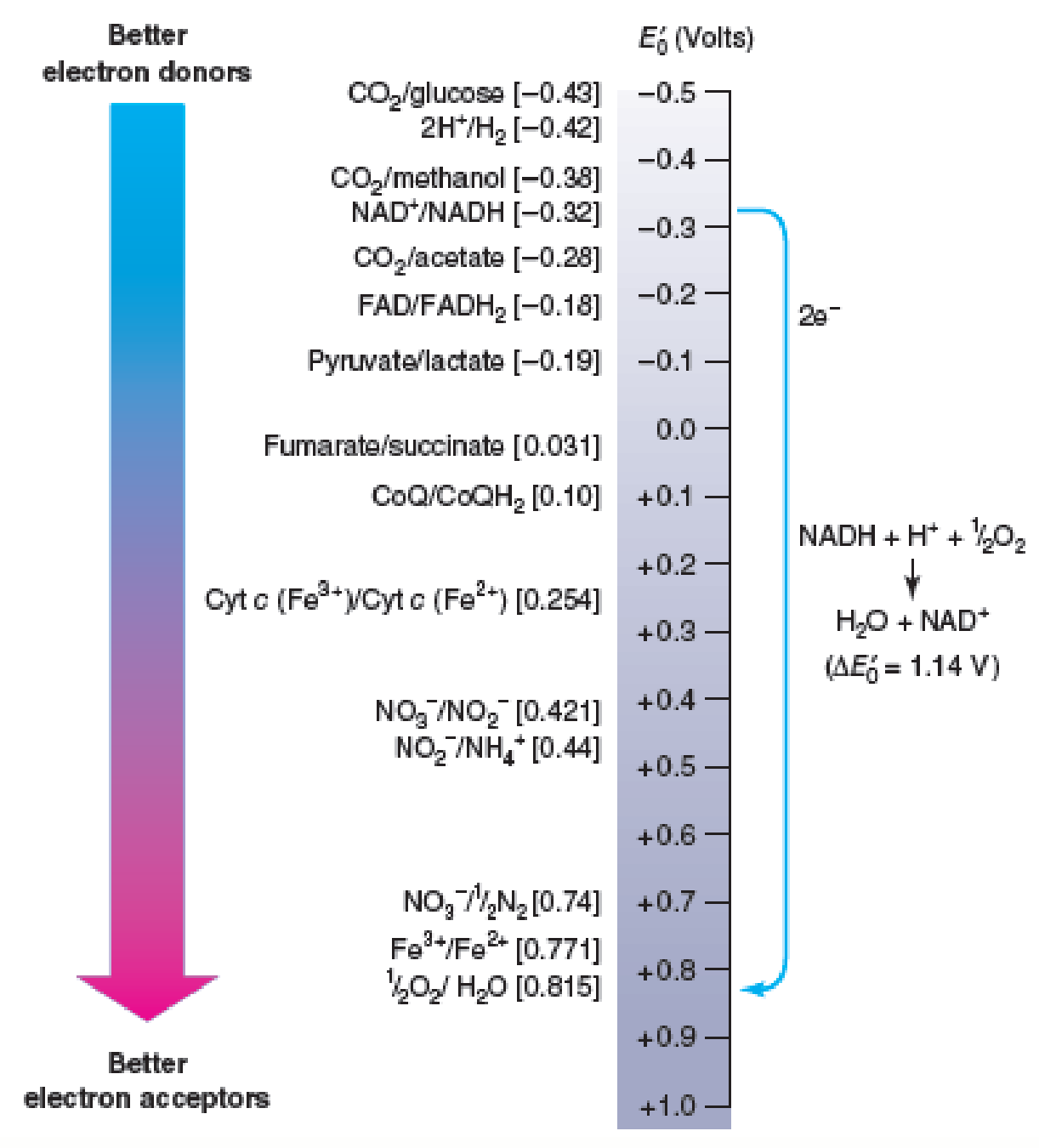
Figure 10.6 Electron Movement and Reduction Potentials. Electrons spontaneously move from donors higher on the tower (more negative potentials) to acceptors lower on the tower (more positive potentials). That is, the donor is always higher on the tower than the acceptor. For example, NADH will donate electrons to oxygen and form water in the process. Some typical conjugate redox pairs are shown on the left, and their reduction potentials are given in brackets.
Refer to figure 10.6 and determine the E′0 for NAD+/NADH and coenzyme Q/CoQH2. Suggest a plausible E′0 value for FMN.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Figure 1 shows the catalytic triad of a-chymotrypsin. Identify A, B and C. Describe the
subsequent steps of stage 1 and stage 2 of a- chymotrypsin mechanism. Illustrate with diagrams.
A
B
CH2
H-N
N: H-O
Figure 1
Activity of the light reactions (ETC) can be assayed using an artificial, reducible compound (e- acceptor) added to take the place of the terminal electron acceptor. The artificial e- acceptor used is called DCPIP, a blue dye that turns colorless when it accepts electrons. The reaction is:
H2O + DCPIPoxidized (blue) --> O2 + DCPIPreduced (colorless)
What molecule from the light reactions of photosynthesis does DCPIP compete with?
NADPH
H2O
ATP
ADP
NADP+
Activity of the light reactions (ETC) can be assayed using an artificial, reducible compound (e- acceptor) added to take the place of the terminal electron acceptor. The artificial e- acceptor used is called DCPIP, a blue dye that turns colorless when it accepts electrons. The reaction is:
H2O + DCPIPoxidized (blue) --> O2 + DCPIPreduced (colorless)
What molecule from the light reactions of photosynthesis does DCPIP compete with?
Chapter 10 Solutions
Prescott's Microbiology
Ch. 10.1 - Figure 10.2 The Relationship of G to the...Ch. 10.1 - Prob. 1CCCh. 10.1 - Prob. 2CCCh. 10.1 - Prob. 3CCCh. 10.1 - Prob. 4CCCh. 10.2 - Why is ATP called a high-energy molecule? How is...Ch. 10.2 - Describe the energy cycle and ATPs role in it....Ch. 10.3 - Prob. 1MICh. 10.3 - Prob. 2MICh. 10.4 - Figure 10.6 Electron Movement and Reduction...
Ch. 10.4 - How is the direction of electron flow between...Ch. 10.4 - When electrons flow from the NAD+/NADH conjugate...Ch. 10.4 - Which among the following would be the best...Ch. 10.4 - In general terms, how is G related to E0? What is...Ch. 10.4 - Name and briefly describe the major electron...Ch. 10.6 - Will an enzyme with a relatively high Km have a...Ch. 10.6 - Prob. 2MICh. 10.6 - Prob. 1CCCh. 10.6 - Prob. 2CCCh. 10.6 - How does enzyme activity change with substrate...Ch. 10.6 - What special properties might an enzyme isolated...Ch. 10.6 - What are competitive and noncompetitive...Ch. 10.6 - How are enzymes and ribozymes similar? How do they...Ch. 10.7 - Figure 10.19 Allosteric Regulation. The structure...Ch. 10.7 - Prob. 2MICh. 10.7 - Define the terms metabolic channeling and...Ch. 10.7 - Define allosteric enzyme and allosteric effector.Ch. 10.7 - Prob. 3CCCh. 10.7 - Prob. 4CCCh. 10.7 - Prob. 5CCCh. 10 - Prob. 1RCCh. 10 - Prob. 2RCCh. 10 - Prob. 3RCCh. 10 - Examine the structures of macromolecules in...Ch. 10 - Examine the branched pathway shown here for the...Ch. 10 - Prob. 3AL
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Activity of the light reactions can be assayed using an artificial, reducible compound (e- acceptor) added to take the place of the natural electron acceptor. The artificial e- acceptor used is called DCPIP, a blue dye that turns colorless when it accepts electrons. The reaction is: H20 + DCPIPoxidized (blue) ->02 + DCPIPreduced (colorless) To detect changes in the color of the DCPIP, a spectrophotometer is used to measure light intensity as a function of wavelength. When light is passed through a solution, the solute may either absorb or scatter light resulting in different color and opacity of the solution. DCPIP absorbs light in its oxidized state but not in its reduced state. Data below shows the results of an experiment using this assay to test the effects of an herbicide on photosynthesis. 0.6 0.55 0.5 No Chloroplasts 0.45 Chloroplasts in the 0.4 dark Chloroplasts in the light 0.35 0.3 10 15 20 Time (minutes) How would you interpret the above data? Results indicate that the…arrow_forwardIdentify the Class and Subclass.arrow_forwardLysozyme is a catalytic enzyme that cleaves NAG-NAM units in the bacte- rial cell wall, and is found in external secretions of the body. Why does thisarrow_forward
- V-class proton pumps run backward relative to the F-class ATP synthase. Consider the cartoon, which shows the conformations of the beta-subunits and ATPIADP + Pj of the F-class synthase. Which of the following associations between the conformation of the beta subunit and ATP/ADP + P¡ is correct for V- Binding Change Mechanism loose binding ADP+P ATP ATP class pumps? C repeat ADP + P, ADP АТР tight binding АТР +P оpen АТР O The open conformation releases ATP. Hydrolysis of ATP to ADP + P¡ drives the change from tight to loose. O Binding of ADP + P¡ drives change from open to loose. Hydrolysis of ATP to ADP + Pj drives the change from open to loose.arrow_forwardWhich of the three graphs below has the largest kcat? Explain. Which of the three graphs below has the tightest subtrate binding? Explain. Which of the three graphs has the largest specificity or efficiency constant? Explain. Not all graphs have to be used as answers. Some graphs may qualify for more than one question. Explain.arrow_forwardExplain why the coupled reaction ATP → ADP + Pi in the P-class ion pump mechanism does not involve direct hydrolysis of the phosphoanhydride bond.arrow_forward
- Below is a plot of Vo vs. [S] for a specific allosteric enzyme under different conditions. Which of the following best describes the graph? 4 (S] O Adding a positive modulator to #2 would result in curve 3. Curve 1 represents maximum inhibition. Line 4 is valid exclusively for curves 1 and 2. Adding a positive modulator to #1 would result in curve 2. O Curve 3 represents the effect of a negative modulator added to curve 2.arrow_forwardA. Lineweaver-Burk plot of the enzyme with increasing amounts of substrate in the absence or the presence of the inhibitor is shown below. Graph A : x-intercept Graph B : x-intercept = - 0.012, y-intercept = 0.8 Graph C : x-intercept = - 0.027, y-intercept = 0.8 Graph D : x-intercept = - 0.039, y-intercept = 0.8 - 0.007, y-intercept = 0.8 Graph A 4 Graph B Graph C Graph D 1 -0,04 -0,02 0,00 0,02 0,04 1/[Substrate] (uM) (i) Which graph indicates an enzymatic reaction without inhibitor? (ii) Which type of inhibitor is it? Briefly explain. (iii) Which graph indicates the highest concentration of inhibitor? (iv) Calculate the Vmax and Km of the graph showing an enzymatic reaction with the lowest concentration of inhibitor. Show the steps of calculation and unit in your answers. Keep 2 decimal places in your answers. 1/Rate (umol/min)arrow_forward(No more than one page). Explain why quaternary structure is necessary for cooperativity and allosteric regulation. Use hemoglobin as an example. Include the various allosteric effectors that influence oxygen binding.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Introduction to the NIOSH Manual of Analytical Methods Fifth edition; Author: Centers for Disease Control and Prevention (CDC);https://www.youtube.com/watch?v=B5rUrKLMoas;License: Standard Youtube License