
Concept explainers
Classify each of the following compounds as a normal
a. 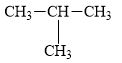
b. 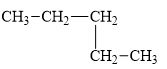
c. 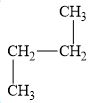
d. 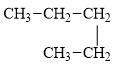
e. 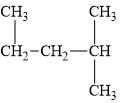
f. 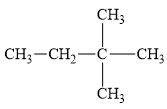
(a)
Interpretation:
The alkane
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound,
Explanation of Solution
The alkane is shown below.
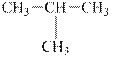
Figure 1
Branched alkanes do not have linear structure and they have alkyl substituents. In the above compound,
The compound,
(b)
Interpretation:
The alkane pentane is to be classified as normal alkane or a branched alkane.
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound, pentane is a normal alkane.
Explanation of Solution
The alkane is shown below.
Figure 2
Normal alkanes have linear structure and have no alkyl substituents attached to it. The above compound, pentane has linear structure and has no alkyl substituents. Therefore, it is a normal alkane.
The compound, pentane is a normal alkane.
(c)
Interpretation:
The alkane butane is to be classified as normal alkane or a branched alkane.
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound, butane is a normal alkane.
Explanation of Solution
The alkane is shown below.
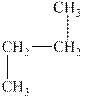
Figure 3
Normal alkanes have linear structure and have no alkyl substituents attached to it. The above compound, butane has linear structure and has no alkyl substituents. Therefore, it is a normal alkane.
The compound, butane is a normal alkane.
(d)
Interpretation:
The alkane pentane is to be classified as normal alkane or a branched alkane.
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound, pentane is a normal alkane.
Explanation of Solution
The alkane is shown below.
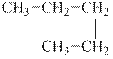
Figure 4
Normal alkanes have linear structure and have no alkyl substituents attached to it. The above compound, pentane has linear structure and has no alkyl substituents. Therefore, it is a normal alkane.
The compound, pentane is a normal alkane.
(e)
Interpretation:
The alkane
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound,
Explanation of Solution
The alkane is shown below.
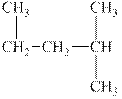
Figure 5
Branched alkanes do not have linear structure and they have alkyl substituents. In above compound,
The compound,
(f)
Interpretation:
The alkane
Concept introduction:
Alkanes are acyclic saturated hydrocarbons. They are also known as the paraffin. The alkanes contain singly bonded carbon atoms and hydrogen atoms. The alkanes have general formula
Answer to Problem 11.28E
The compound,
Explanation of Solution
The alkane is shown below.
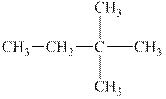
Figure 6
Branched alkanes do not have linear structure and they have alkyl substituents. In the above compound,
The compound,
Want to see more full solutions like this?
Chapter 11 Solutions
CHEMISTRY FOR TODAY+OWLV2 24 MO>IP<
- Why are different conformations of an alkane not considered structural isomers?arrow_forwardFor each of the following alcohols, give the systematic name and specify whether the alcohol is primary, secondary, or tertiary. a. b. c.arrow_forwardThe general formula of an alkane is CnH2n+2 . What is the general formula of an (a) alkene? (b) alkyne? (c) alcohol derived from an alkane?arrow_forward
- a) what two types of bonds are present in alkynes but not alkanes? b) draw the structure of an alkyne that would not have one of the two bonds. Explain.arrow_forwardConsider the following isomers of hexene: A. a disubstituted cis- alkene B. the geometric isomer of ISO C. a tri-substituted Z- alkene D. most stable alkene E. has the highest boiling point F.arrow_forwardDraw the following organic molecules using the linear structurearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





