
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
2nd Edition
ISBN: 9780077633707
Author: Janice Smith
Publisher: Mcgraw-hill Higher Education (us)
expand_more
expand_more
format_list_bulleted
Question
Chapter 11, Problem 11.30UKC
Interpretation Introduction
Interpretation:
The polymerized product of the given compound has to be determined.
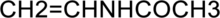
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the class of polymer for each structure.
Doct
Structure A:
Structure A is a:
O polycarbonate
polyester
O polyurethane
O polyamide
Fotoy
Structure B:
Structure B is a:
CH3
O polycarbonate
O polyurethane
polyamide
O polyester
Part A
What are glycosides, and how can they be formed?
A glycoside is the acetal product that results from rerction of the hemiacetal -OH group of a carbohydrate with an aldehyd
O A glycoside is the acetal product that results from reaction of the hemiacetal-OH group of a carbohydrate with an alcohol.
A glycoside is the hemiacetal product that results from reaction of the acetal-OH group of a carbohydrate with an alcohol.
Submit
Previous Answers Request Answer
Figure 1 shows the chemical structure of maleic acid and adipic acid. A polyester is formed when
the maleic anhydride is hydrolysed to form maleic acid and react with the ethylene glycol. Styrene
is then added and polymerized.
HO-
(i)
||
||
-C-CH=CH-C-OH
Maleic acid
i
HO-C-(CH₂)4-C-OH
Adipic acid
||
Figure 1: Chemical structure of maleic acid and adipic acid
Write down the reaction equation and indicate the type of polymerization reactions
involved and describe the chemical composition of the polymer produced by drawing the
partial structure of the resulting polymer.
(ii) Distinguish between the polymer formed using maleic anhydride and ethylene glycol and
the one in which the maleic anhydride was replaced with adipic acid with the aid of a
reaction equation between adipic acid and ethylene glycol.
Chapter 11 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
Ch. 11.1 - Convert each condensed structure to a complete...Ch. 11.1 - Determine whether each molecular formula...Ch. 11.1 - Give the molecular formula for each of the...Ch. 11.2 - Give the IUPAC name for each alkene. a. (CH3CH2)2C...Ch. 11.2 - Prob. 11.5PCh. 11.2 - Give the structure corresponding to each name. a....Ch. 11.3 - Prob. 11.7PCh. 11.3 - Bombykol is secreted by the female silkworm moth...Ch. 11.3 - Prob. 11.9PCh. 11.3 - Prob. 11.10P
Ch. 11.3 - Prob. 11.11PCh. 11.5 - Prob. 11.12PCh. 11.5 - Prob. 11.13PCh. 11.6 - Prob. 11.14PCh. 11.6 - Prob. 11.15PCh. 11.7 - Prob. 11.16PCh. 11.7 - Prob. 11.17PCh. 11.9 - Prob. 11.18PCh. 11.9 - Draw the structure corresponding to each name. a....Ch. 11.10 - Prob. 11.20PCh. 11.10 - Prob. 11.21PCh. 11.10 - Prob. 11.22PCh. 11 - Prob. 11.23UKCCh. 11 - Prob. 11.24UKCCh. 11 - Prob. 11.25UKCCh. 11 - Prob. 11.26UKCCh. 11 - Answer the following questions about compound A,...Ch. 11 - Prob. 11.28UKCCh. 11 - Prob. 11.29UKCCh. 11 - Prob. 11.30UKCCh. 11 - Prob. 11.31UKCCh. 11 - Prob. 11.32UKCCh. 11 - Prob. 11.33APCh. 11 - Prob. 11.34APCh. 11 - Prob. 11.35APCh. 11 - Prob. 11.36APCh. 11 - Prob. 11.37APCh. 11 - Falcarinol is a natural pesticide found in carrots...Ch. 11 - Prob. 11.39APCh. 11 - Prob. 11.40APCh. 11 - Prob. 11.41APCh. 11 - Prob. 11.42APCh. 11 - Prob. 11.43APCh. 11 - Give the structure corresponding to each IUPAC...Ch. 11 - Leukotriene C4 is a key compound that causes the...Ch. 11 - Prob. 11.46APCh. 11 - Prob. 11.47APCh. 11 - Prob. 11.48APCh. 11 - Prob. 11.49APCh. 11 - Prob. 11.50APCh. 11 - Prob. 11.51APCh. 11 - Prob. 11.52APCh. 11 - Prob. 11.53APCh. 11 - Prob. 11.54APCh. 11 - Prob. 11.55APCh. 11 - Prob. 11.56APCh. 11 - Prob. 11.57APCh. 11 - Draw the products formed in each reaction.Ch. 11 - Prob. 11.59APCh. 11 - Prob. 11.60APCh. 11 - Prob. 11.61APCh. 11 - Prob. 11.62APCh. 11 - Prob. 11.63APCh. 11 - Prob. 11.64APCh. 11 - Prob. 11.65APCh. 11 - Prob. 11.66APCh. 11 - Prob. 11.67APCh. 11 - Prob. 11.68APCh. 11 - Prob. 11.69APCh. 11 - Prob. 11.70APCh. 11 - Prob. 11.71APCh. 11 - Prob. 11.72APCh. 11 - Prob. 11.73APCh. 11 - Prob. 11.74APCh. 11 - Prob. 11.75APCh. 11 - Prob. 11.76APCh. 11 - Prob. 11.77APCh. 11 - Prob. 11.78APCh. 11 - Prob. 11.79APCh. 11 - Prob. 11.80APCh. 11 - Prob. 11.81APCh. 11 - Prob. 11.82APCh. 11 - Prob. 11.83APCh. 11 - Prob. 11.84APCh. 11 - Prob. 11.85APCh. 11 - Prob. 11.86APCh. 11 - Are cis-2-hexene and trans-3-hexene constitutional...Ch. 11 - Prob. 11.88CP
Knowledge Booster
Similar questions
- + Mannose is a monosaccharide involved in the glycosylation of certain proteins. Several genetic disorders are associated with enzymes involved in mannose metabolism. Monosaccharides form a new functional group when the straight chain sugar cyclizes. What is the type of functional group formed in mannose after it undergoes cyclization to form the furanose ring? HO OH OH 0- OH OH A) aldehyde B) ketone C) hemi-acetal D) ether E) esterarrow_forwardIUPAC name? (CH3)2C=CHCH2CH2OHarrow_forwardThe following chemical reaction is used to synthesize a flavouring agent that has an aroma similar to bananas. H₂SO4(aq) CH3COOH(1) + CH₂(CH₂)₂OH(1) I || Identify the type of reaction that is represented by this synthesis. Select one: O hydrogenation O esterification O addition O substitution O elimination Identify the functional group and the IUPAC name for each of the three compounds in the reaction below: H₂SO4(aq) I Compound Functional Group IUPAC Name II CH₂COOH(1) + CH₂(CH₂)₂OH(1) I || III CH3COO(CH₂)₂CH₂(1) + H₂O(1) III IV ◆ → ◆ CH3COO(CH₂)₂CH₂(1) + H₂O(1) IV ||| ◆ ◆arrow_forward
- 12 Draw the structure of polymer formed using the following monomer. `CH=CH2 Give the reagent that can initiate the polymerizationarrow_forwardDraw structures for the following compunds:arrow_forwardLactose contains what number of anomeric carbons and what number of glycosidic bonds: CH2OH ОН CH2OH ОН OH OH OHarrow_forward
- Provide the polymer showing 5 repeating units for the following alkene: CH2CH=CH2CH3arrow_forwardExplain the effects the addition of glycerol (a triol) will have on the molecular architecture of the following polymerization reactions: a) Condensation polymerization of terephthalic acid + ethylene glycol b) Condensation polymerization of 6-hydroxyhexanoic acidarrow_forwardWhat polymer is formed in each of the following reactions? Draw each polymer in polymer notation.n →> Catalyst Part B CH₂ Catalyst n CH H* 12D EXP. CONT. Marvin JS by ChemAxon A # H с N S CI Br - PFarrow_forward
- The following reaction is an example of a reaction. нн H H Heat or light H-C-c-H + Cl-ci н—с—с—сі: + н—сi: нн нн ethane chloroethane O Substitution Elimination O Hydrogenation O Chlorinationarrow_forwardWhat alcohol is formed when the alkene CH3CH2CH=CH2 is treated with H2O in the presence of H2SO4?arrow_forwardCH2CH3 H3C–C–F + CH;O-arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning